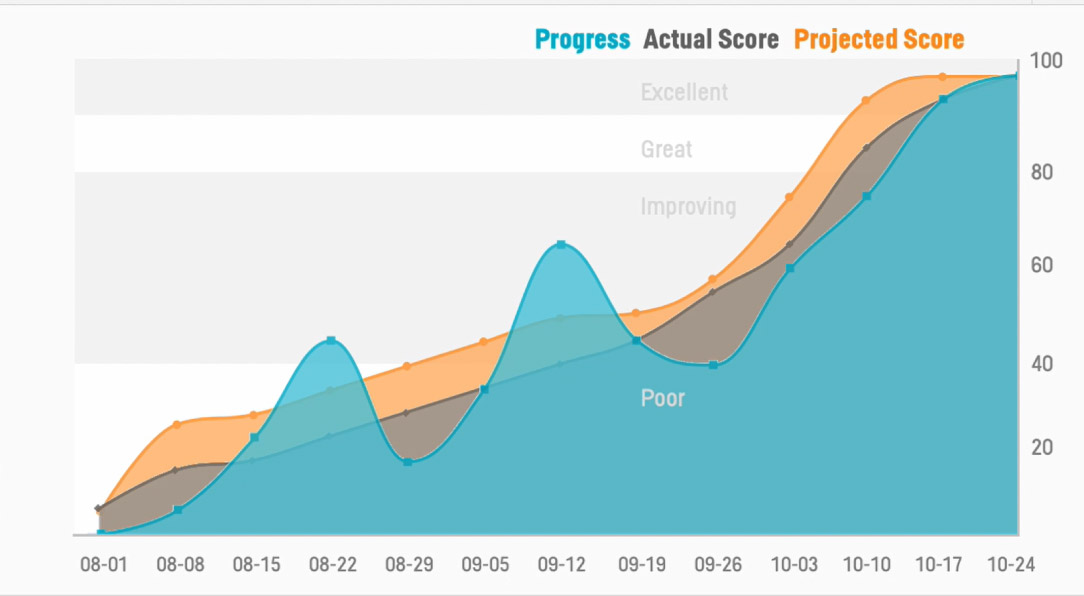
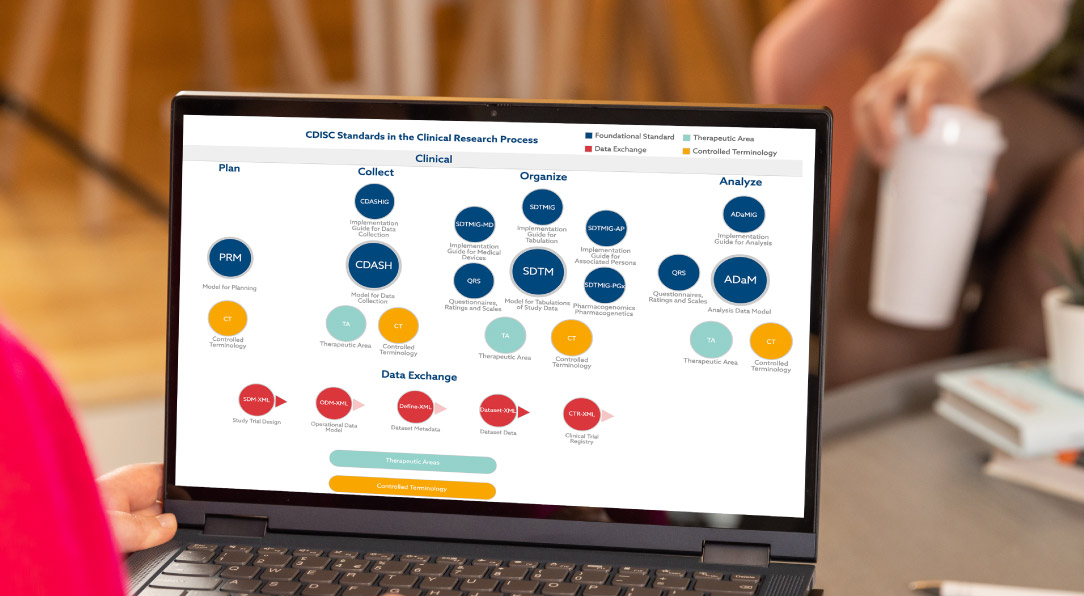
Pinnacle 21® clinical data standardization software empowers clinical teams to standardize, validate, and harmonize data effortlessly. Achieve flawless submissions and accelerate development with confidence.
Pinnacle 21®
Achieve submission ready conformance with Pinnacle 21 clinical data standardization software

Learn more about Pinnacle 21 clinical data standardization software
Optimize quality & speed from setup through submission with clinical data standardization
Manage and optimize your clinical trial data from the start.
Why implement clinical data standardization software?
The Pinnacle 21 clinical data management software platform transforms clinical data into submission-ready deliverables – enhancing data quality across studies, and validating datasets against global regulatory standards. By integrating metadata management and continuous data validation in a collaborative platform, Pinnacle 21 speeds timelines, reduces risk, and ensures data integrity throughout the clinical data flow.
Metadata management for eCRFs and submissions.
Centralize metadata for eCRFs, data transfers and submission datasets, and harmonize in line with clinical data standards, such as CDISC, as well as your own in-house standards.
Real-time data validation with issue tracking.
Collaborate with CROs, developers, and others in a central workspace to resolve issues discovered during validation.
Effortless collaboration across both internal and external clinical teams.
Design your transfer specifications with each vendor and accept data packages in the cloud.
Leaders in clinical data management
Pinnacle 21 Enterprise Software Suite
With Pinnacle 21 Enterprise as your foundation, add additional functionality according to your requirements. Get in touch to find out more about adding additional modules to your Enterprise platform.
Continuous data validation
Pinnacle software suite
Pinnacle 21 Community resources
Support from our technical experts
Need a hand preparing your SDTM datasets or submission deliverables? Our technical support team combines expert knowledge of clinical data standards with decades of experience helping thousands of clients achieve regulatory compliance and expedited submissions.
Data standardization and validation resources
View all
Aligned with CDISC data standards
The Clinical Data Interchange Standards Consortium (CDISC) develops data standards. Many regulatory agencies require drug submissions to conform to CDISC data standards. Pinnacle 21 embraces all CDISC guidance.

With Pinnacle 21, your data is secure
Certara holds ISO 27001 certification for Certara’s Information Security Management System (ISMS). We have implemented robust security controls, undergone rigorous risk assessments, and continuously strive for improvement. Pinnacle 21 ensures full compliance with global data protection standards, offering peace of mind for sensitive analysis.
Transform clinical data with Pinnacle 21
Schedule a consultation to see how Pinnacle 21 clinical data management software can accelerate your path from study design to submission.
FAQs
Who should use Pinnacle 21 Enterprise in a clinical data workflow?
Pinnacle 21 Enterprise is designed for clinical data management teams, statistical programming teams, and regulatory teams responsible for study data standardization and submission. The platform supports cross-functional collaboration by providing a centralized platform for study setup, SDTM mapping specs, continuous validation, issue management, metadata governance, and submission deliverables. This makes it especially valuable for organizations handling multiple clinical programs.
How does Pinnacle 21 Enterprise support regulatory compliance for clinical submissions?
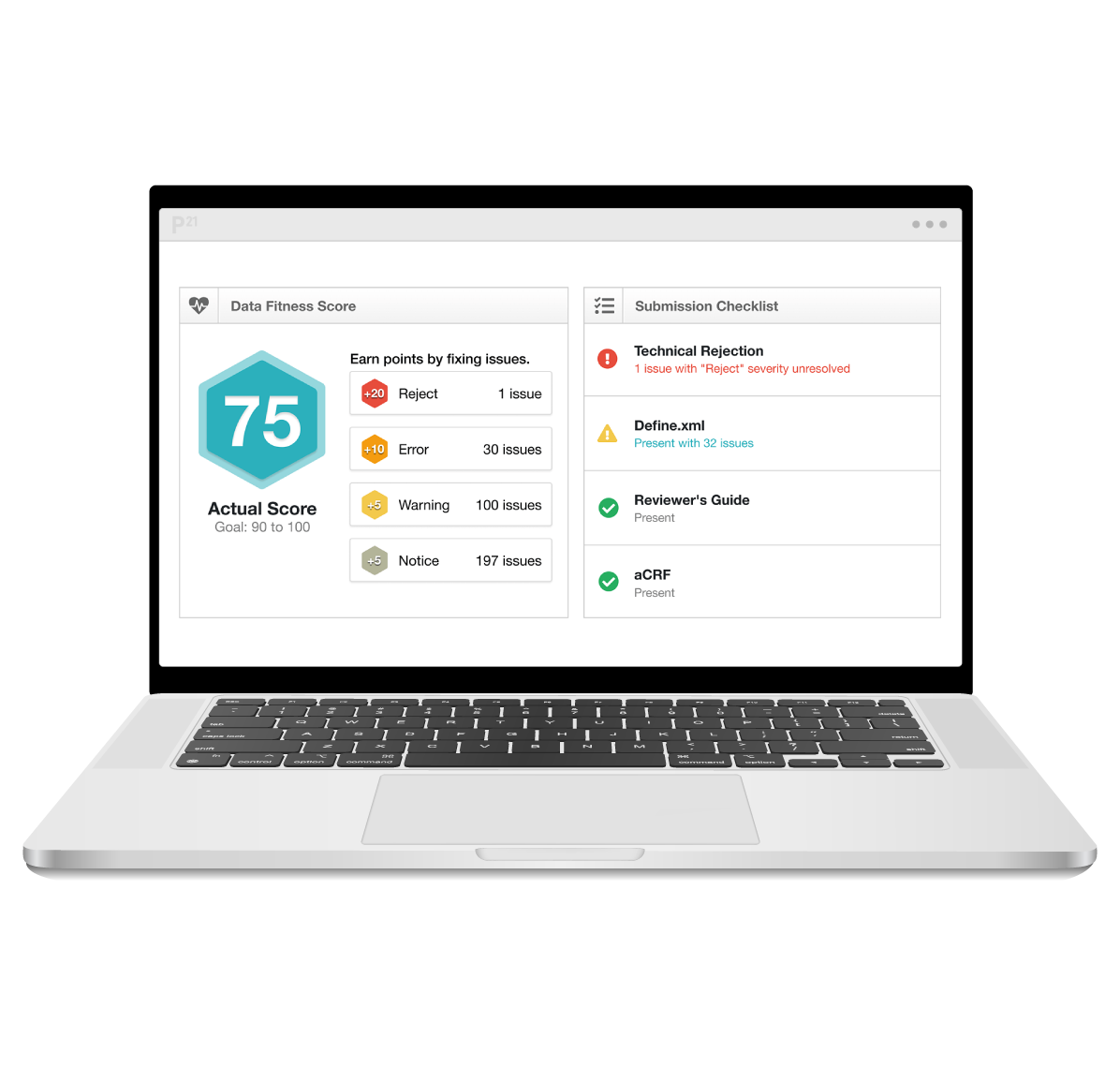
Pinnacle 21 Enterprise supports regulatory compliance by validating clinical datasets against the latest CDISC standards and global regulatory rules. Validation engines are maintained in line with updates, and ‘data fitness’ scores guide users towards compliance. Issues are flagged based on regulatory impact, allowing teams to focus on fixing those issues that pose the greatest risk to submission. Teams are empowered with the knowledge and guidance to provide high-quality, compliant, submission-ready data packages.
What can you do with the advanced Pinnacle 21 Enterprise platform that you can't do with the free Community tool?
There are many differences between the advanced and free versions of Pinnacle 21. Key differences center around compliance, submission readiness, issue management, submission deliverables, governance and study oversight. Read this blog for a point-by-point comparison of the two platforms: How does Pinnacle 21 Enterprise differ from the Community version?
When is the right time to upgrade from Pinnacle 21 Community to Enterprise?
Deciding this often depends on the number and complexity of studies being managed, as well as the study phase. As studies advance, especially from Phase II to Phase III, clinical data volumes grow and complexity increases, making it harder to reconcile issues and generate high quality deliverables at scale. Read this blog to understand more about transitioning from Community to Enterprise: Clinical trial data validation: When to transition from P21 Community to Enterprise
Where can I download Pinnacle 21 community?
If you’re new to Pinnacle 21 and want to get a feel for its basic validation capabilities, you can access Community here: https://help.pinnacle21.certara.net/en/articles/9736610-download-pinnacle-21-community