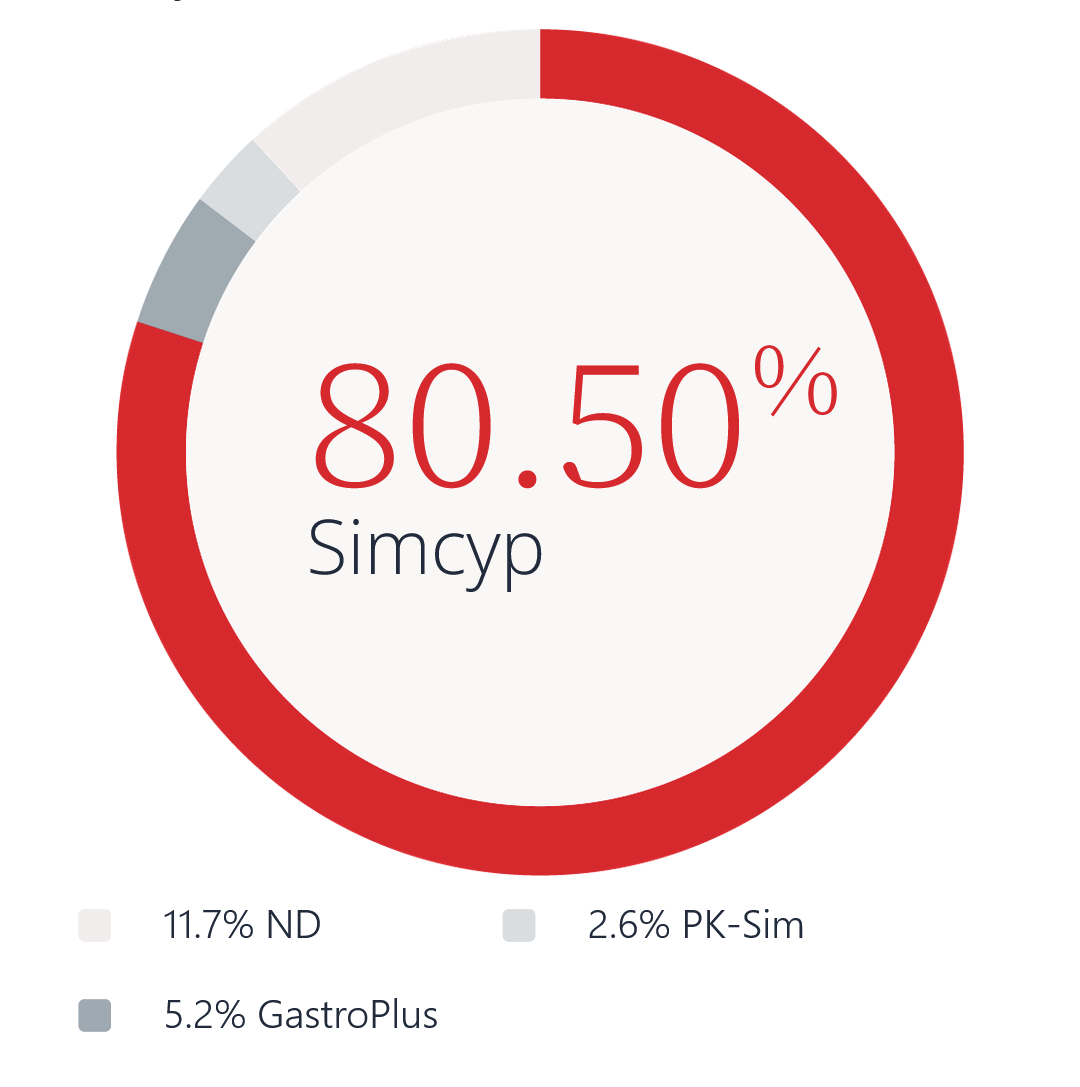
PBPK modeling is a mechanistic modeling approach increasingly adopted across all stages of drug development. Simcyp® provides a seamless PBPK modeling environment for all stages of drug development, reducing the reliance on extensive in vivo studies.
As the most widely adopted platform for PBPK modeling in drug development, Simcyp is the only technology solution suite developed in collaboration with 37 of the top global pharmaceutical companies over the last 25 years. A testament to its leadership in PBPK, Simcyp is licensed by 11 regulatory agencies worldwide and over 120 novel drugs approved by the FDA used Simcyp in lieu of clinical studies.