March 31, 2026
CROs can deliver compliant datasets and documentation faster, supporting shorter submission timelines for sponsors.
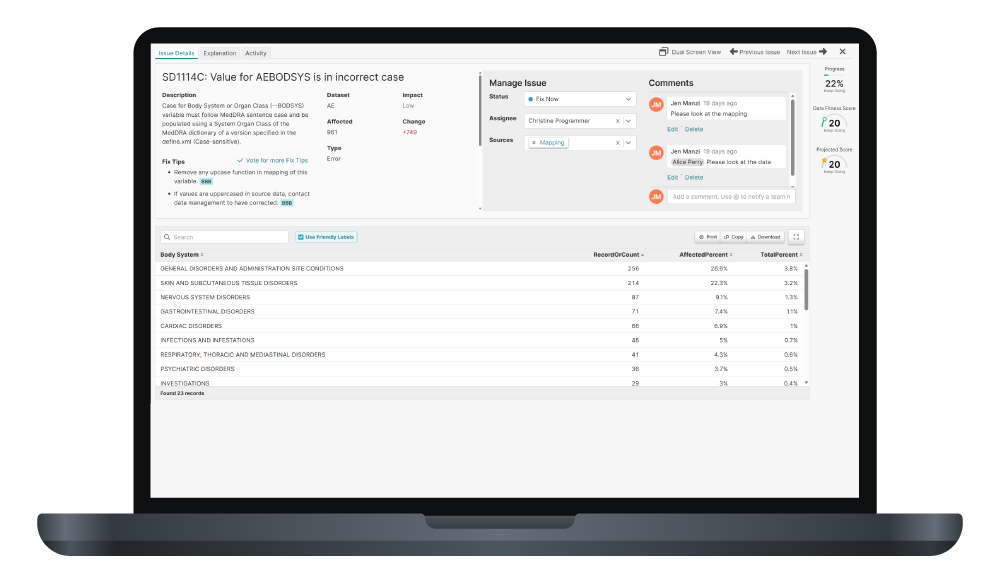
By proactively identifying and resolving issues upstream, CROs can position sponsors for more confident and successful submissions.
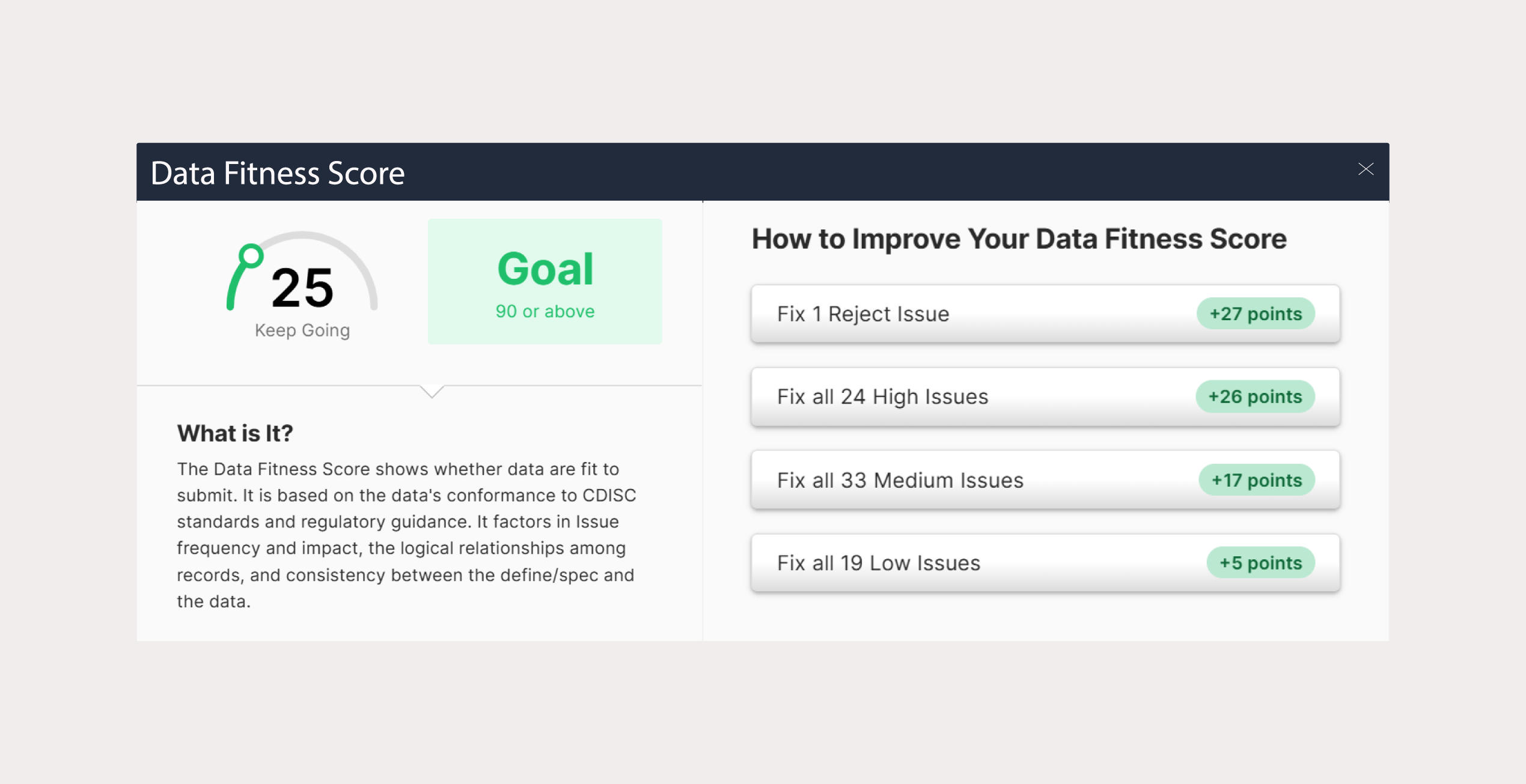
This level of transparency strengthens sponsor trust and streamlines review cycles, improving overall service quality.
By boosting efficiency, CROs can take on more work and optimize margins, while maintaining high-quality sponsor outputs.
By minimizing the risk of failed submissions, CROs deliver not just data, but confidence.
Learn more about how CROs can effectively manage submission complexity at scale with Pinnacle 21 Enterprise.


Jen Manzi
Subject Matter Expert and User Advocate, Pinnacle 21 by CertaraJen Manzi is a Subject Matter Expert and User Advocate at Pinnacle 21. She has over 20 years of Pharma/Life Sciences industry experience in Clinical Trials and Safety Data Management. Jen has held various roles within these areas, including eCRF Programmer, SDTM Delivery Lead, Product Owner and Programmer of Batch Processes, Vendor Relationship Manager, Program and Process Improvement Manager, and Validation Lead.
Make an inquiry about Pinnacle 21 Enterprise
Pinnacle 21 Enterprise builds on Community’s core validation capabilities with:
Make an inquiry to learn how your organization can benefit from reduced risk, increased quality, and guided submission readiness with Pinnacle 21 Enterprise.
FAQs
What are platforms for collaboration between sponsors and CROs?
These platforms centralize workflows, such as validation, issue management, and cross-team communication, providing greater visibility and improved efficiency across the study lifecycle.
How to improve sponsor CRO collaboration?
Purpose built platforms for collaboration between sponsors and CROs facilitate efficient teamworking and dramatically strengthen partnerships. Pinnacle 21 Enterprise provides advanced CRO sponsor collaboration tools: sponsors and CROs work from a secure, shared environment, with dashboards, audit trails, and issue tracking, which removes the reliance on manual spreadsheets, complex email chains and unaudited offline collaboration.
Why is validation assurance important for CROs?
Validation assurance is an important key deliverable for CROs. They’re accountable for providing sponsor data that meets regulatory requirements, in order to reduce the risk of failed submissions. Regulatory submission readiness software for CROs, like Pinnacle 21 Enterprise, gives CROs the precise tools for achieving approval outcomes for clients.
How do CROs ensure submission readiness?
CROs must ensure submission readiness by adhering to CDISC standards, controlled terminology, and agency specific rules. Using sponsor and CRO collaboration platforms ensures a secure, central environment, built on the latest CDISC aligned regulatory framework, with continuous validation for optimal submission readiness.
What tools do CROs use for regulatory submissions?
Some organizations use tools such as Pinnacle 21 Community for basic validation requirements. However, in order to deliver accurate, timely submission deliverables at scale, regulatory submission readiness software for CROs, such as Pinnacle 21 Enterprise, is a more fit-for-purpose solution. The platform equips CROs with the tools to provide submission assurance for every sponsor, when juggling priorities across multiple clients and studies. Hear how Summit Analytical are using Enterprise to optimize regulatory submissions >>
Can Pinnacle 21 Enterprise help CROs scale operations?
Yes. Such platforms for collaboration between sponsors and CROs provide a scalable infrastructure, with audit tracking, traceability, and integrated automation of validation processes and issue management. CROs have the tools to effectively manage multiple sponsors and studies simultaneously, in a secure, cloud-based environment.





