April 29, 2026
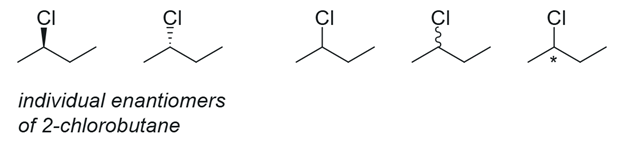
Figure 1. Graphical depictions of 2-chlorobutane. Depending on the author and reader, the meaning may be or may not be interpreted as intended.
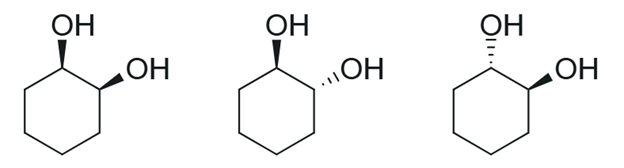
Figure 2. Stereoisomers of cyclohexane-1,2-diol.
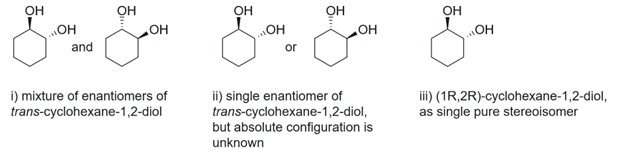
Figure 3. Unambiguous depictions of trans-cyclohexane-1,2-diol.
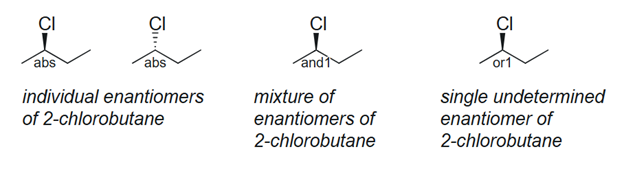
Figure 4. Enhanced stereochemical labels distinguish stereopure compounds, racemic mixtures, and unknown stereochemistry.
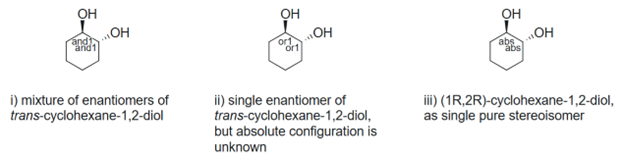
Figure 5. Enhanced stereochemical labels distinguish enantiomers, diasteromers, and stereopure compounds.
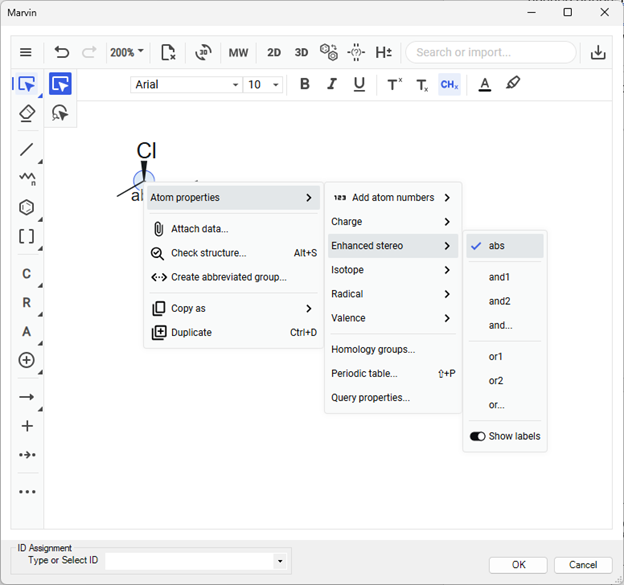
Figure 6. Setting enhanced stereolabels in Marvin.
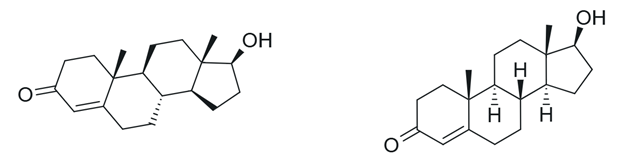
Figure 7. Depictions of testosterone.
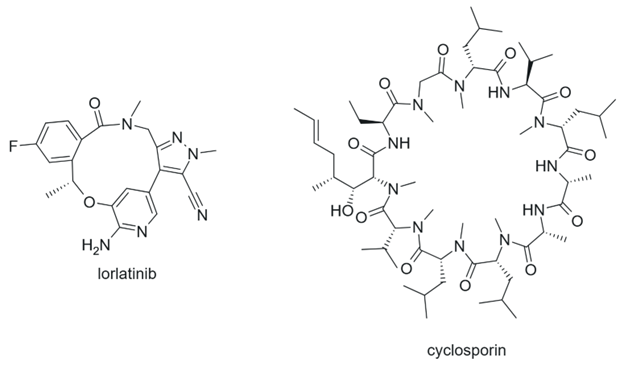
Figure 8. Macrocycles.
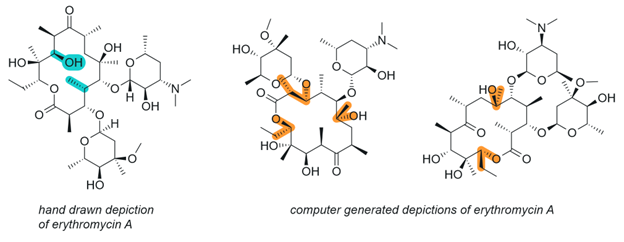
Figure 9. Hand drawn and computer generated depictions of erythromycin A.
References
1. Graphical Representation of Stereochemical Configuration (IUPAC Recommendations 2006), https://iupac.qmul.ac.uk/drawing/stereo.html

Jeremiah Malerich, PhD
Solution ConsultantDr. Jeremiah Malerich is a Solution Consultant working in the Discovery Portfolio of Certara. He joined Chemaxon – acquired by Certara in 2025 – two years ago after a long career in the CRO business supporting drug discovery programs. In his previous life, he saw how critical robust cheminformatic tools were to the success of programs, and he now brings that experience to Certara and its partners. His background is synthetic organic chemistry with a PhD from the University of California, Berkeley.
Book a Demo
Reach out to us
– Leave your contact information
– Summarize what you are looking for
– Our colleagues will get back to you soon





