Certara is dedicated to transforming drug discovery and development for good. We harness the power of biosimulation, advanced analytics, scientific, strategic and regulatory expertise to create a future where treatments reach patients faster and more efficiently.
Certara is transforming drug discovery and development for good
Welcome to Certara. We transform drug development for the benefit of humans. Our commitment is to empower innovators, researchers, and regulatory experts with software and service solutions tailored exactly to meet their needs, fostering success across the entire drug development lifecycle.
Certara overview
Certara Mission & Vision
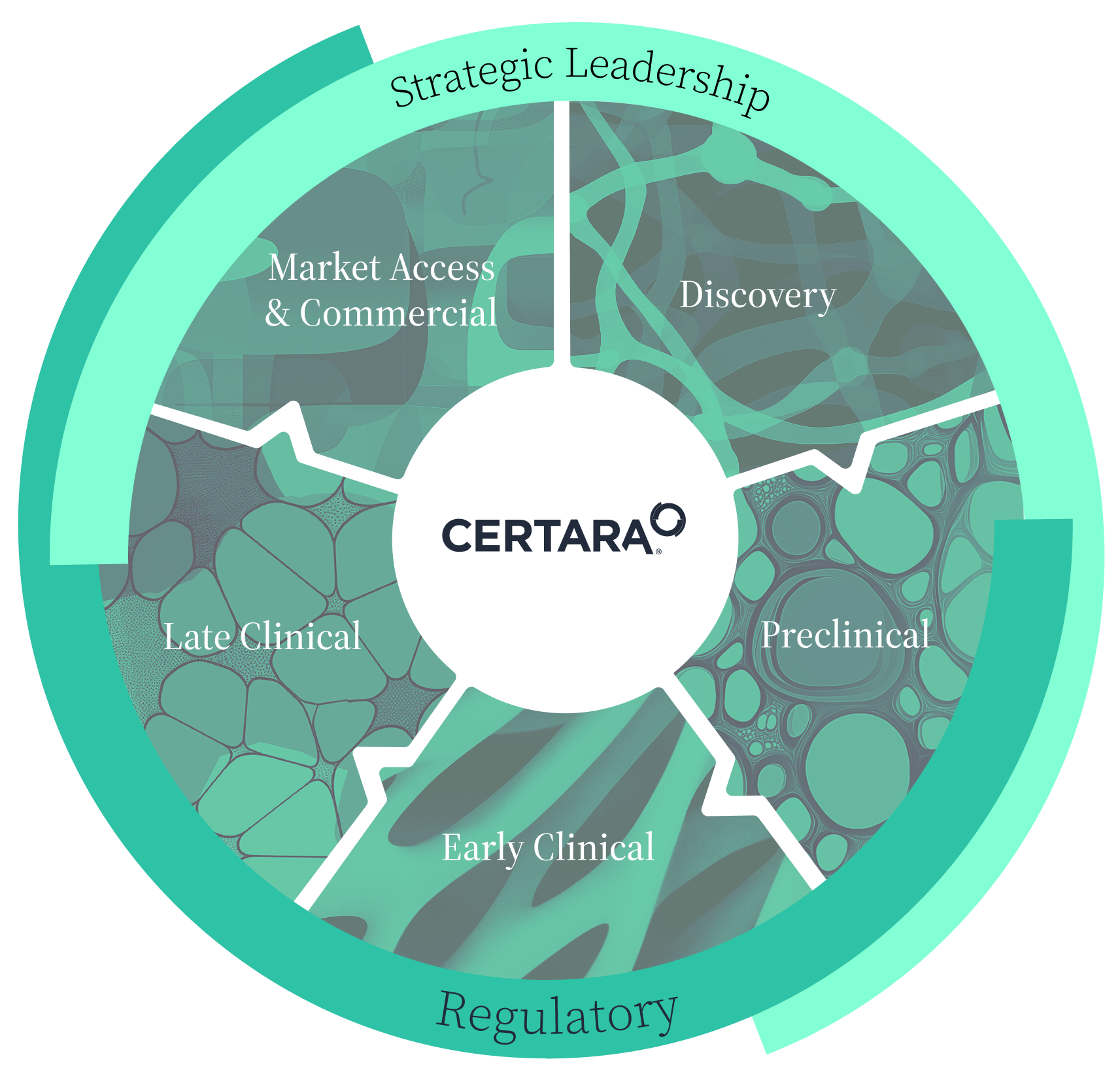
Client-centric solutions that improve drug development
From molecule to market, we tailor solutions to meet our clients’ most pressing challenges. Through strategic leadership, scientific and regulatory rigor along with advanced predictive technologies, Certara provides comprehensive solutions to optimize drug development processes, reduce risks, and improve outcomes.
The keenest minds in life science and technology
With over 350 scientific consultants and industry experts, Certara’s voice in drug development is respected around the world. Our customers are located in over 70 countries.
Leaders in the science of life
Over millenia, nature dictated that you lost 5 of 7 children and died before you reached 50. Certara plays in one of the most important sectors: the application of science and technology to improve and extend human health and life.
Regulatory adoption
Certara works closely with global regulatory agencies to ensure compliance and streamline submission processes, accelerating submissions and enhancing patient access to better medicines and treatments.
Meet our leadership
Our leadership team brings unparalleled expertise and vision that drives Certara’s mission. With decades of experience in science, technology, and healthcare, we’re united in our goal to transform the drug development landscape.
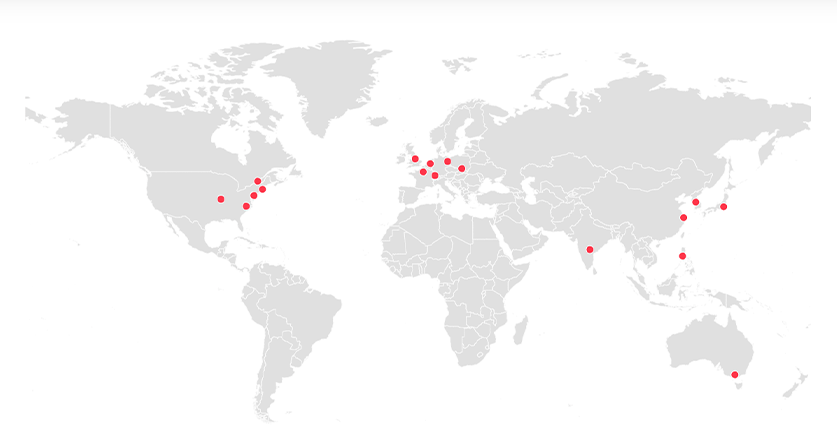
Global presence
Our footprint extends across the globe, partnering with industry leaders and regulatory agencies to advance therapeutic innovation. From regulatory adoption to strategic partnerships, we’re at the heart of the scientific community.
Continuous investment in innovation
In 2008, Tripos International and Pharsight Corporation came together to form Certara. Since then, we have achieved incredible growth through acquisitions and the pharmaceutical industry and regulatory agencies’ adoption of model-informed drug development (MIDD).
Today, we are the global leader in biosimulation.
Certara’s responsibility
We’re committed to sustainable practices, ethical business, and making a positive impact on global health. Our responsibility extends to patients, partners, and the planet, ensuring that every solution we deliver aligns with our core values.





Discover more about Certara
Strategic partnership
Collaboration is key to our success. Certara’s network of partners spans academia, industry, and government, driving innovation and expanding possibilities in drug development.
Investing in Innovation
Certara is at the forefront of transforming drug development, delivering exceptional value to stakeholders and shaping the future of healthcare. Explore how our strategic vision and innovative solutions drive sustainable growth and success.
Shape the Future of Drug Development
Certara is recognized as an ethical, inclusive and responsible employer. You’ll find inspiring and engaged people at our workplaces. Do you want to be one of us? We invite you to apply.



