
Genentech was developing polatuzumab vedotin (Polivy), an anti-CD79b-vc-monomethyl auristatin E (MMAE) antibody-drug conjugate (ADC) to treat patients with transplantation-ineligible relapsed/refractory (R/R) diffuse large B-cell lymphoma (DLBCL). MMAE is a potent anti-cancer drug that is a CYP3A substrate and a weak competitive CYP3A inhibitor.
The cytotoxic payload of an ADC is a small molecule that could be metabolized and cleared via metabolic enzymes (cytochrome P450 enzymes (CYPs), UGTs, drug transporters, etc). Concomitant medications that alter inhibit or induce CYP3A could alter the pharmacokinetics (PK) of unconjugated MMAE and thereby affect clinical outcomes. Thus, the Genentech team needed to assess the risk of CYP3A-mediated DDIs for polatuzumab vedotin.
The results of simulations performed using the model were used to inform the drug label without the need for dedicated clinical trials.
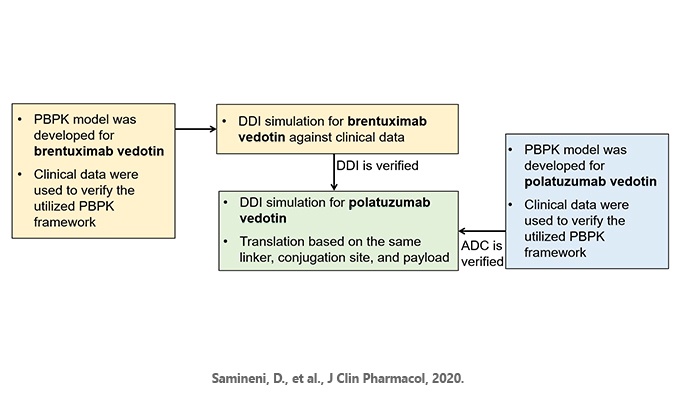
Genentech scientists used the Simcyp Simulator to develop a physiologically-based pharmacokinetic (PBPK) model to perform an in silico DDI study. The model was first developed and verified using data from an existing clinical DDI study for brentuximab vedotin, an ADC using the same linker-drug combination as polatuzumab vedotin. Simulations performed using this model predicted that polatuzumab vedotin would not significantly inhibit or induce CYP3A. The PBPK simulations also demonstrate limited impact of strong CYP3A inhibitors/inducers on the exposure of MMAE.
Contact us




