April 16, 2026
Looking to go beyond the basics of PK parameters? Enhance your knowledge with comprehensive insights from distinguished experts in pharmacokinetics and pharmacodynamics.
Looking to go beyond the basics of PK parameters?
Authored by Prof. Johan Gabrielsson, the trusted reference book Pharmacokinetic and Pharmacodynamic Data Analysis provides comprehensive insights into pharmacokinetics, pharmacodynamics, and PK/PD concepts.
Register with your professional or academic email to access the full standard edition – completely free.
This blog was originally published in March 2013 and has been updated for accuracy.

Ana Henry
Executive Director, Training & Certara UniversityAna leads the Certara University team in providing modeling and simulation for new drug development through education, skills, and expertise in the global healthcare industry. Ana has more than 20 years experience in a variety of roles in the industry. She has extensive experience in pharmaceutical training, software demonstration, software support, and product management, Ana is also an adjunct faculty member at Skaggs College of Pharmacy and Pharmaceutical Sciences at the University of Colorado.
Phoenix WinNonlin: A trusted software tool for analyzing bioequivalence and bioavailability studies
As the industry standard for NCA, PK/PD studies, and toxicokinetic (TK) modeling, Phoenix WinNonlin delivers 30 years of reliability and innovation. Trusted by regulatory agencies like the FDA, PMDA, CFDA, and MHRA, it ensures compliance, reduces manual effort, and enhances productivity.

FAQs
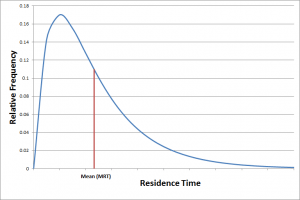
What is the difference between mean residence time (MRT) and half-life?
MRT represents the average time a drug molecule stays in the body, while half-life measures how long it takes for half of the drug concentration to be eliminated. MRT considers the full distribution of molecule lifetimes, whereas half-life focuses only on the rate of decline, making MRT more comprehensive in certain pharmacokinetic analyses.
How does poor PK sampling in the terminal elimination phase of the drug affect MRT accuracy?
Inadequate sampling during the terminal elimination phase can significantly distort MRT calculations. Since MRT relies on accurate estimation of the drug’s elimination tail, missing or sparse late-time data can lead to under- or overestimation, making the results unreliable for interpretation or decision-making.
What is the relationship between MRT and drug clearance?
MRT and clearance are inversely related when the volume of distribution is held constant. In linear pharmacokinetic systems, MRT can be expressed as Vss/CL, linking residence time in the body to both drug distribution and elimination. Specifically, MRT can be expressed as the ratio of volume of distribution to clearance in certain models. This relationship helps contextualize MRT within broader pharmacokinetic behavior, linking how long a drug stays in the body to how efficiently it is removed.
Schedule a Phoenix demo
Ready to See Phoenix in Action? Transform your PK/PD analyses with a guided demo tailored to your needs. We’ll show you how Phoenix can bridge your journey across drug development.






