Drug clearance is an extremely important topic in the science of pharmacokinetics. Drug clearance defines how much drug should be administered, how frequently to dose a patient, and how two interacting drugs will affect a patient. The primary PK parameter clearance is very similar to its friend, volume of distribution. Clearance (CL) is a proportionality factor that relates the concentration of drug measured in the body to the rate of elimination. In mathematical terms:
Rate of Elimination = CL * C(t)
In most cases, CL is considered to be constant (Red line). This means that the rate of elimination (Green line) changes in direct proportion to the concentration of drug measured (Blue line). This concept is illustrated in the following graph:
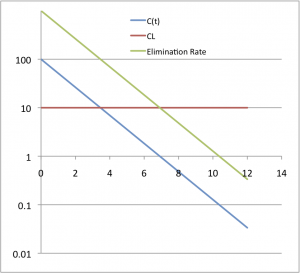
Some might ask, what does it mean to “clear” a drug. Clearance is a collection of processes by which the body removed the drug from the body. This occurs in two general ways.
Metabolism – Drug can be metabolized, or converted into other chemical species.
Elimination – Drug is removed from the body by a specific organ (e.g. kidneys)
When either metabolism or elimination occur, the drug is no longer available to cause a pharmacological effect in the body. Metabolism often takes place in the liver. The liver is full of enzymes such as the cytochrome P450s which convert the drug into more hydrophobic hydrophilic species which can then be eliminated. Elimination occurs when the drug is removed from the blood or plasma and placed “outside” of the body. The kidney is an elimination organ. The kidneys take drugs from the blood and move them to the urine.
In some cases, the clearance of the drug changes with the concentration of drug measured in the body. This situation is called non-linear clearance, or non-linear pharmacokinetics. Often this occurs when elimination or metabolism is controlled by a single enzymatic pathway that can get saturated.
In summary, clearance is a proportionality factor, just like volume of distribution. Clearance relates the rate of elimination to the concentration measured in the body. Clearance is a function of organ function, and efficiency and is different for each drug.
Many physiological changes are associated with obesity and can potentially impact on pharmacokinetics. This can require adjustments to be made to the standard doses for normal weight patients in order to ensure safety and efficacy of drug therapy. Read this case study to learn how physiologically-based pharmacokinetic (PBPK) models incorporate the known, relevant demographic, anatomical and physiological variables associated with obesity to predict drug clearance with reasonable accuracy in obese and morbidly obese subjects.


