March 17, 2026
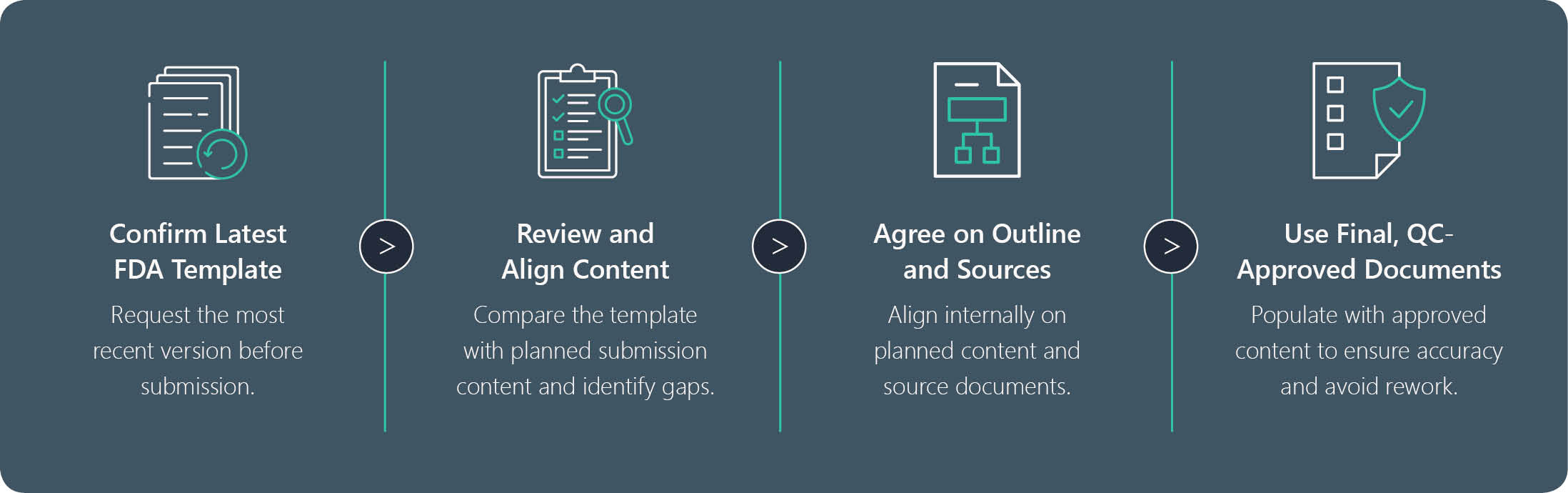

Explore how dependency mapping helps teams plan, author, and maintain submission content while minimizing errors and delays.

Karen Randolph
Karen Randolph has 20 years of experience in the pharmaceutical industry. She leads multifunctional teams in developing strategies and documentation for orphan drug designation applications and marketing applications for approval of orphan drugs in the United States and other geographies such as the European Union.
References
FDA Oncology Center of Excellence. 2018 Annual Report. https://cdn.cancerhistoryproject.com/media/2023/03/03144018/oncology-center-excellence-2018-annual-report-04.10.19_3.pdf. Accessed 18 December 2025.
FDA Oncology Center of Excellence. 2019 Annual Report. https://cdn.cancerhistoryproject.com/media/2023/03/06145827/OCE-2019-Annual-Report_508_V3.pdf. Accessed 18 December 2025.
FDA Oncology Center of Excellence. 2020 Annual Report. https://cdn.cancerhistoryproject.com/media/2023/03/06145827/OCE-2019-Annual-Report_508_V3.pdf. Accessed 18 December 2025.
FDA Oncology Center of Excellence. Oncology Regulatory Review (2021 Annual Report). https://www.fda.gov/about-fda/2021-oce-annual-report/oncology-regulatory-review. Accessed 18 December 2025.
FDA Oncology Center of Excellence. Oncology Regulatory Review (2022 Annual Report). https://www.fda.gov/about-fda/2022-oce-annual-report/oncology-regulatory-review. Accessed 18 December 2025.
FDA Oncology Center of Excellence. Oncology Regulatory Review 2023. https://www.fda.gov/about-fda/2023-oce-annual-report/oncology-regulatory-review-2023. Accessed 18 December 2025.
FDA Oncology Center of Excellence. Oncology Regulatory Review 2024. https://www.fda.gov/about-fda/2024-oce-annual-report/oncology-regulatory-review-2024. Accessed 18 December 2025.
FDA Oncology Center of Excellence. Assessment Aid. https://www.fda.gov/about-fda/oncology-center-excellence/assessment-aid. Accessed 18 December 2025.
FDA Oncology Center of Excellence. Real Time Oncology Review Pilot Program. https://www.fda.gov/about-fda/oncology-center-excellence/real-time-oncology-review. Accessed 18 December 2025.
This blog was originally published in July 2021 and has been updated for accuracy.
FAQs
Is the FDA Assessment Aid required for oncology NDA or BLA submissions?
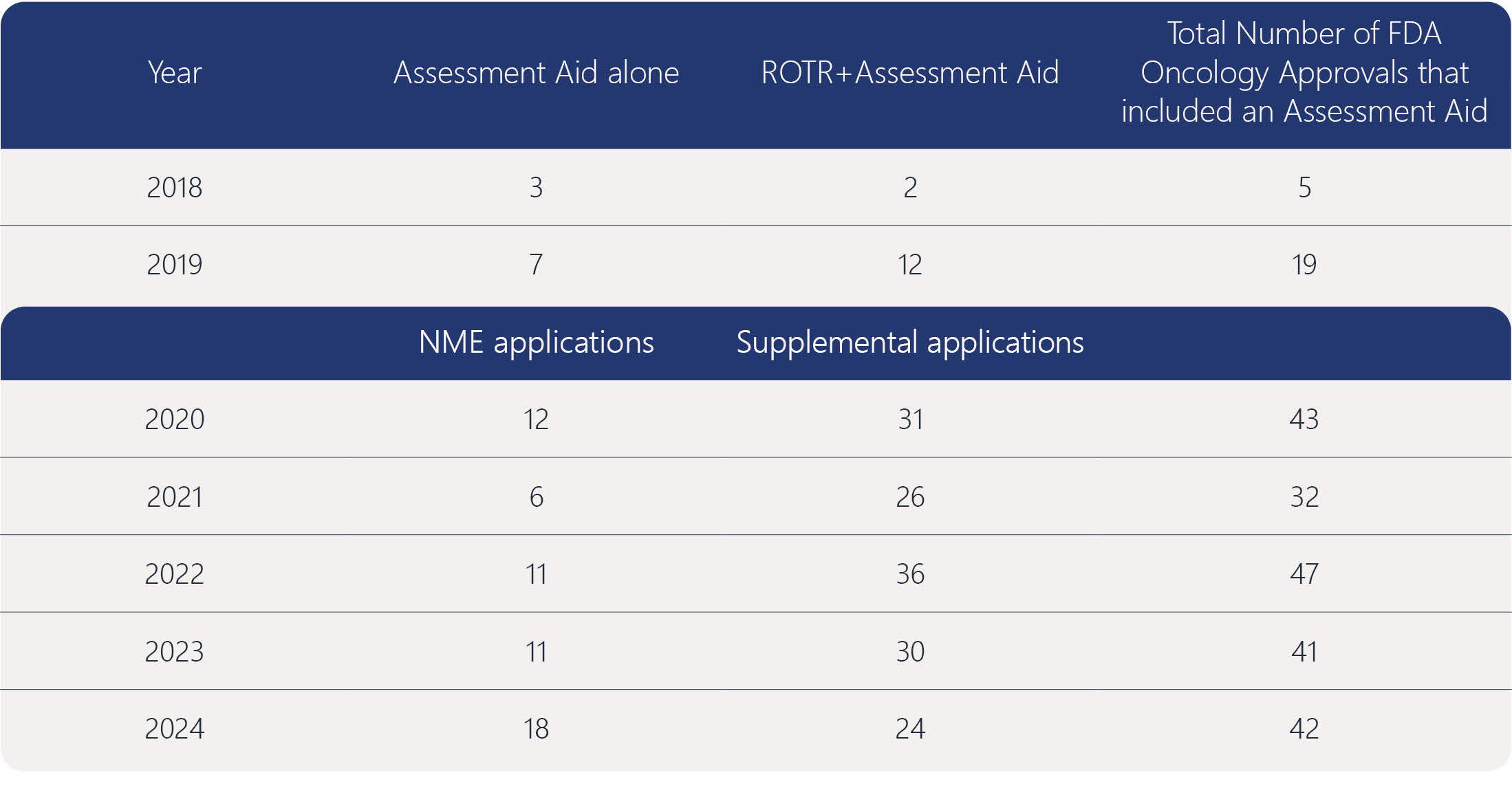
No, the FDA Oncology Assessment Aid is voluntary and not required for NDA or BLA submissions. However, the FDA prefers its inclusion for applications submitted under the Real-Time Oncology Review (RTOR) program. Participation has increased significantly since 2018, reflecting its value in facilitating oncology application reviews.
What information is included in an FDA Oncology Assessment Aid?
The Assessment Aid uses a standardized template covering nonclinical data, clinical pharmacology, efficacy, and safety. For supplemental applications without new data, sponsors may indicate that no additional information is included. The document summarizes key evidence from the submission and should not introduce new data.
Does using an Assessment Aid improve FDA review timelines?
The Assessment Aid is designed to streamline FDA review by focusing on critical data and analysis rather than recapitulating information. While it does not guarantee faster approval, it can support review efficiency, particularly in oncology submissions and RTOR pathways.
Let’s transform your regulatory journey