I apologize to all my readers for such a long lapse between posts. After a very busy summer and fall, I am back to posting regularly to my blog about PK/PD topics.
When analyzing PK/PD data, one of the most important plots used to visualize the data is to plot time-matched PK/PD data on a scatter plot. The X-axis has the PK concentration and the Y-axis has the PD data. Two examples of these scatter plots are shown below.
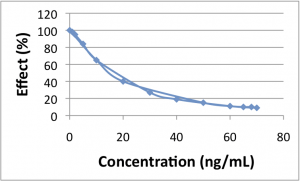
No Hysteresis
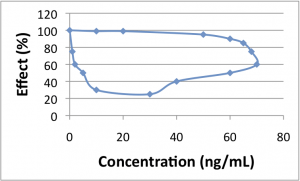
Hysteresis
The first plot shows a relationship with no hysteresis, and the second shows hysteresis. The easiest way to identify hysteresis is by drawing a vertical line on the concentration-effect plot. If that line crosses the curve in 2 places, indicating 2 different response levels for a single drug concentration, then you have hysteresis. In the first plot (no hysteresis) a vertical line at 40 ng/mL corresponds to a single effect level (20%). However, in the second plot (hysteresis) a vertical line at 40 ng/mL corresponds to both effect levels of 40% and 100%.
A hysteresis is neither good nor bad when reviewing PK/PD data. A hysteresis loop simply means that there is a time delay between the measured concentration and the effect response. Normally this means that the measured effect is indirectly affected by the measured concentration. To properly model this relationship, you would want to use an effect compartment or an indirect PK/PD model.
So a hysteresis loop simply provides information on how to model your PK/PD data.
The methods used to characterize the pharmacokinetics (PK) and pharmacodynamics (PD) of a compound can be inherently complex and sophisticated. PK/PD analysis is a science that requires a mathematical and statistical background, combined with an understanding of biology, pharmacology, and physiology. PK/PD analysis guides critical decisions in drug development, such as optimizing the dose, frequency and duration of exposure, so getting these decisions right is paramount. Selecting the tools for making such decisions is equally important. Fortunately, PK/PD analysis software has evolved greatly in recent years, allowing users to focus on analysis, as opposed to algorithms and programming languages. Read our white paper to learn about the key considerations when selecting software for PK/PD analysis.


