Radiprodil is a selective allosteric modulator of the NR2B N-methyl-D-aspartate receptor GluN2B-NMDA that was being investigated to treat infantile spasm syndrome. Alongside clinical trials, a radiprodil physiologically-based pharmacokinetic-pharmacodynamic (PBPK-PD) model was built based on in vitro data in the Simcyp Simulator. The model was expanded to incorporate our understanding of the developmental physiology and ontogeny of elimination, to predict PK in the age-appropriate pediatric population for infantile spasm syndrome (aged 2 to 14 months). We then linked the PBPK model to a receptor occupancy (RO) PD model in the Simcyp Simulator.
In vitro experiments based on hepatocyte data were used to further assess our understanding of the ontogeny, first for CYP1A2/acetaminophen and then for radiprodil. From those results, which strongly suggested a lack of ontogeny effect, a dosing decision tree was developed, recommending a low RO and low dose escalation approach. To our knowledge, this is the first time a PBPK model linked to RO has been used to guide dose selection and escalation in the live phase of a pediatric clinical trial.
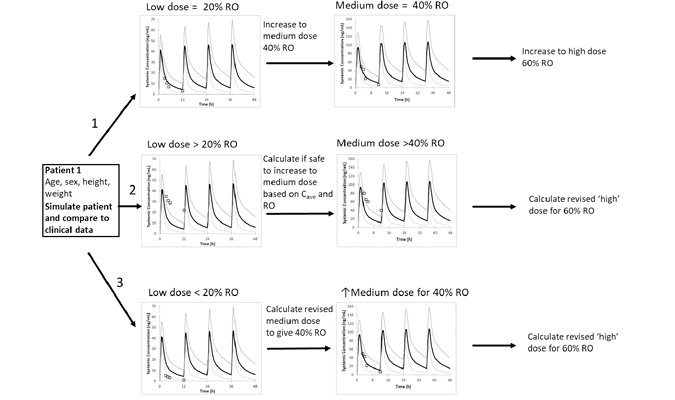
Because the disease is rare and potentially life threatening, there was the need for trial doses to be efficacious from the start, while paying attention to limiting the upper level of RO to avoid unwanted potential side effects associated with high levels of NMDA receptor antagonism. In this case, the use of a PBPK-PD model allowed a potentially more robust dosing strategy to be developed compared to simple allometry alone, which tends to overestimate doses in neonates and infants. The final Simcyp model was used to determine initial doses with age and to guide dose escalation for three individual subjects in the live phase of a clinical trial. Predicted and observed data were in close agreement suggesting that the model was appropriate and adequate for dose selection.
Contact us




