On August 16, 2022, President Biden signed the Inflation Reduction Act of 2022 (IRA) into law. One key objective of the IRA is to shift healthcare costs from patients and the Centers for Medicare & Medicaid Services (CMS) to health insurers and pharmaceutical manufacturers. While the provisions allowing CMS to negotiate drug prices for select Part D (prescription) and Part B (provider-administered) drugs have received the most attention, the impacts of the Part D redesign will be felt earlier and more broadly by patients and payers.
An examination of the impact and likely actions of Part D redesign on managed care payers
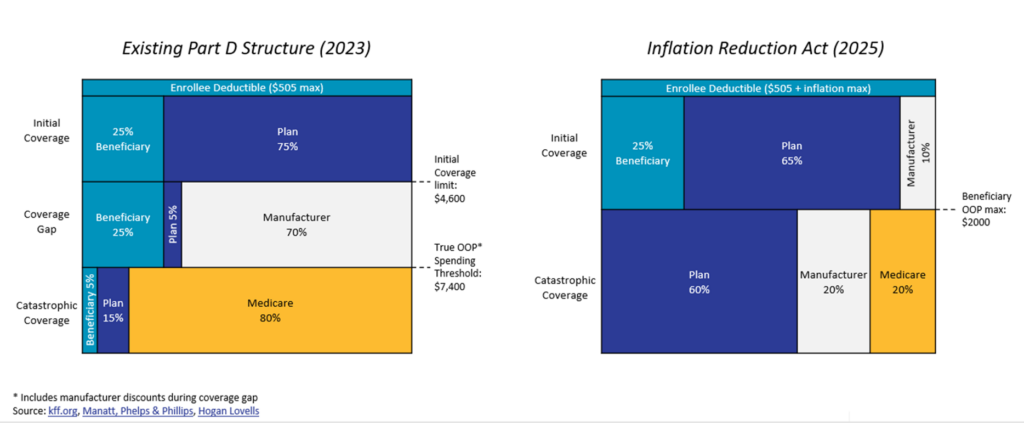
The key elements and implementation dates of the Part D redesign, which apply to both Medicare Advantage-Prescription Drug (MA-PDs) and standalone Prescription Drug Plans (PDPs), are as follows:
- Limiting Medicare Part D premium growth to no more than 6% per year (2024-2030)
- Eliminating the 5% patient coinsurance for Part D catastrophic coverage (2024)
- Setting a $2000 OOP cap in Part D, subject to annual inflationary increases (2025)
- Eliminating the coverage gap, such that Part D beneficiaries, after meeting the deductible, proceed through an initial coverage phase followed by a catastrophic phase (2025)
- Replacing the current “coverage gap” program with a new manufacturer “pay to play” program and a CMS reinsurance policy (2025)
- Manufacturers participating in Part D must offer a 10% discount during the initial coverage period and a 20% discount during the catastrophic coverage period.
- CMS’s share of drug costs during the catastrophic coverage period falls from 80% to either 20% (for brand name drugs) or to 40% (for generics)
- Part D plans’ responsibility during the catastrophic coverage period increases to 60% from 15%
The clear winners here are Medicare patients. Perhaps it is not a coincidence that the most impactful components of President Biden’s signature legislative achievement will be implemented in an election year.
Rising costs for Part D payers
While both the federal government and drug manufacturers will incur new financial responsibilities from the changes, Part D payers are also facing significant new costs:
- Greater use of Part D drugs: Reduced patient cost-sharing of Part D drugs is expected to lead to higher utilization.
- More beneficiaries reaching catastrophic coverage: The catastrophic phase now begins when patients’ out-of-pocket costs reach $2,000. In this phase, enrollees pay nothing.
- Greater plan financial responsibility once a patient reaches catastrophic coverage: Part D plans’ responsibility during the catastrophic coverage period quadruples from 15% to 60%
Compounding this challenge, plans are curtailed from some traditional solutions due to the interplay of new and existing Medicare provisions:
- Limits on premium increases: The 6% annual cap curtails plans’ ability to pass costs to patients; in fact, the average total Part D beneficiary premium is projected to decrease by 1.8% in 2024
- Limited ability to manage some of the highest-cost drugs: CMS requires Part D plans to cover “all or substantially all drugs” in six “protected” classes: anticonvulsants, antidepressants, antipsychotics, antiretrovirals, immunosuppressants, and antineoplastics (which includes many oral chemotherapy drugs). CMS strongly discourages use of step therapy, preferred tiers, or prior authorization for these therapies.[1]
What’s a plan to do?
In the face of these challenges, managed care organizations and the marketplace will have to adapt. This will likely play out in several ways:
- Ramped-up “traditional” utilization management: Plans’ greater incentives to control drug spending will likely lead them to increase the use of such utilization management tools as step therapy and prior authorizations. PDPs, which will not benefit financially from lower medical costs attributable to better adherence to drug therapy, may implement stricter higher utilization management than MA-PDs, which also cover medical benefits.
- Soft utilization management of traditionally unmanaged categories: Payers may become more creative to limit use in traditionally unmanaged categories with high likelihood of pushing the enrollee into catastrophic coverage.
- Incentivization to use lower net price drugs: As Adam Fein has written about, today, Part D plans have two incentives to promote use of higher-list price product over equivalent drugs with lower list prices. First, plans have only 15% financial liability for patients in the catastrophic coverage phase (which is calculated on pre-rebate price). Second, coinsurance tied to list prices leads to cost-related non-adherence. With the IRA’s cap on patient OOP costs and increased financial liability for plans, this strategy becomes less attractive.
- More aggressive contracting with manufacturers: Tighter formulary control by payers will likely force manufacturers to offer to higher discretionary rebates to PBMs and plans in Part D to achieve or maintain access.
Finally….
- Market consolidation: Some MA-PDs and PDPs without sufficient populations to spread the financial impact of the changes may exit the market or merge with other plans.
The IRA’s changes to the Medicare Part D benefit design are undoubtedly going to change the environment in which health insurers and pharma manufacturers interact with each other, as each try to shift their increased cost burden elsewhere.
To learn more about the impact of the IRA, please watch the on demand webinar we gave on this topic.
[1] Step therapy is a process by which payers require patients to take one or more alternative medications before they can access the medicine in question. Tiers are levels of coverage based on the type or usage of the medication. Each tier will have a defined out-of-pocket cost that the patient must pay before receiving the drug. Payers may create “preferred” tiers for branded drugs with lower out-of-pocket costs than branded drugs on the “non-preferred” tier. Prior authorization means that a provider must obtain approval from the payer before the patient can receive the drug.


