May 1, 2026

Why Performing Key CMC Activities Early Can Aid De-risking Your Drug Development Program
This white paper focuses on best practices for developing a CMC strategy for orally administered small molecule drugs.

Deven Shah, BPharm, PhD
VP, Chemistry, Manufacturing, & ControlsDr. Shah leads Certara’s CMC team with over two decades of experience in formulation development, drug delivery, and regulatory interactions.
This blog was originally published in July 2014 and has been updated for accuracy.
Learn more about our Early Development offering
Expert Support at Every Stage from Candidate Selection through Phase 1

FAQs
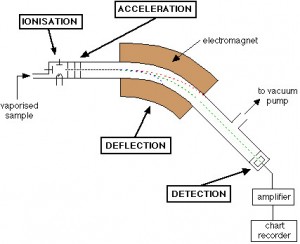
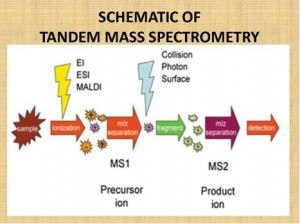
How is LC/MS/MS used in drug development?
Here are some applications of LC/MS/MS in developing a new therapy.
- Pharmacokinetics (PK) & ADME: LC-MS/MS is essential for assessing absorption, distribution, metabolism, and excretion (ADME) profiles of investigational drugs.
- Bioanalysis and Regulatory Compliance: It offers high-throughput, quantitative analysis of drug candidates in biological fluids under GLP (Good Laboratory Practice) guidelines.
- Biomarker Discovery & Validation: LC/MS/MS is used to identify and validate novel biomarkers to measure therapeutic responses in patients.
- Metabolite Profiling & Drug Metabolism: Studies identify active drug metabolites and potential toxicological risks early in development.
- Immunogenicity Testing: Immunogenicity (IG) is defined by the FDA as the propensity of the therapy to generate immune responses to itself and to related therapies or to induce immunologically-related adverse clinical events. LC-MS/MS can be used to detect anti-drug antibodies (ADA) for biopharmaceuticals, thus aiding formulation strategies.
How do Certara’s experts support bioanalytical assay development?
We support our clients throughout the lifecycle of their projects for bioanalytical assay development of diverse modalities encompassing small molecules, peptides, mAbs, bispecific antibodies, oligonucleotides, antibody-drug conjugates (ADCs), and cell therapies. We help with CRO selection as well as oversight of the bioanalytical method development & validation, sample analysis protocols, and analytical report reviews. We provide bioanalytical assay development support for both preclinical and clinical stages and ensure that a phase-appropriate strategy is implemented for relevant PK/PD biomarkers and immunogenicity evaluation. We also provide support for regulatory strategy and compiling required regulatory writing documentation such as the Integrated Summary of Immunogenicity (ISI).
What happens if a program neglects their clinical PK/PD/ADA assay development?
Not developing suitable bioanalytical assays (e.g. pharmacokinetics (PK), anti-drug antibody/neutralizing antibody (ADA/nAb), pharmacodynamic (PD) Biomarkers, and Immune Response Functional Assays) in a timely manner has huge implications on the product’s PK, efficacy, safety, and regulatory acceptability. If robust validated assays aren’t available on time, there is a risk of not being able to determine PK for enabling dose escalation, deriving robust PK-PD modeling, understanding immunogenicity risks, or conducting comparability evaluation for any implemented changes. This in turn can require repeating non-clinical or clinical studies, leading to significant delays to the project timelines (~12-18 months or more) and incurring significantly higher costs. This also carries the risk of not meeting the regulatory standards for the bioanalytical data package resulting in increased scrutiny by the regulatory agencies for the IND/NDA/BLA submissions. There could be major review questions delaying the approvals or resulting in clinical hold.
Contact us our Early Development experts
You May Also Like