9 out of 10 drugs in development fail, costing billions annually, with late-stage failures being the most expensive due to prior investments. A key factor in these failures is sub-optimal trial design.
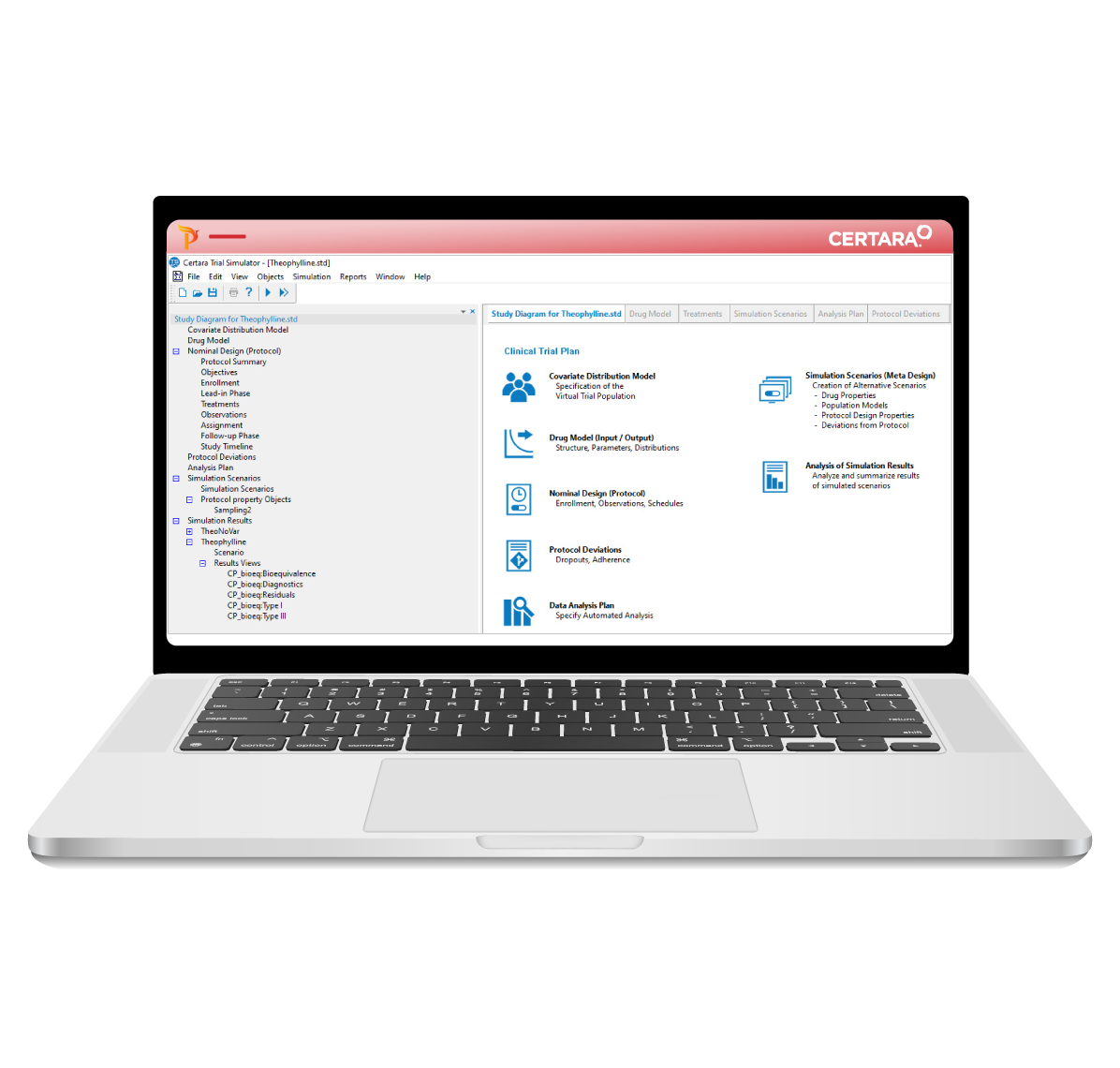
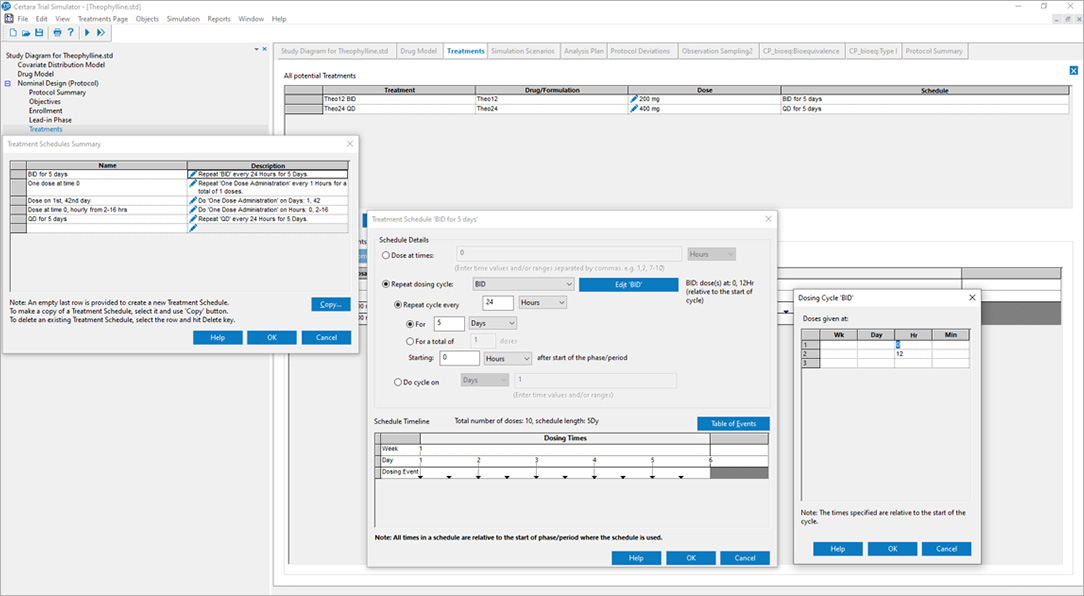
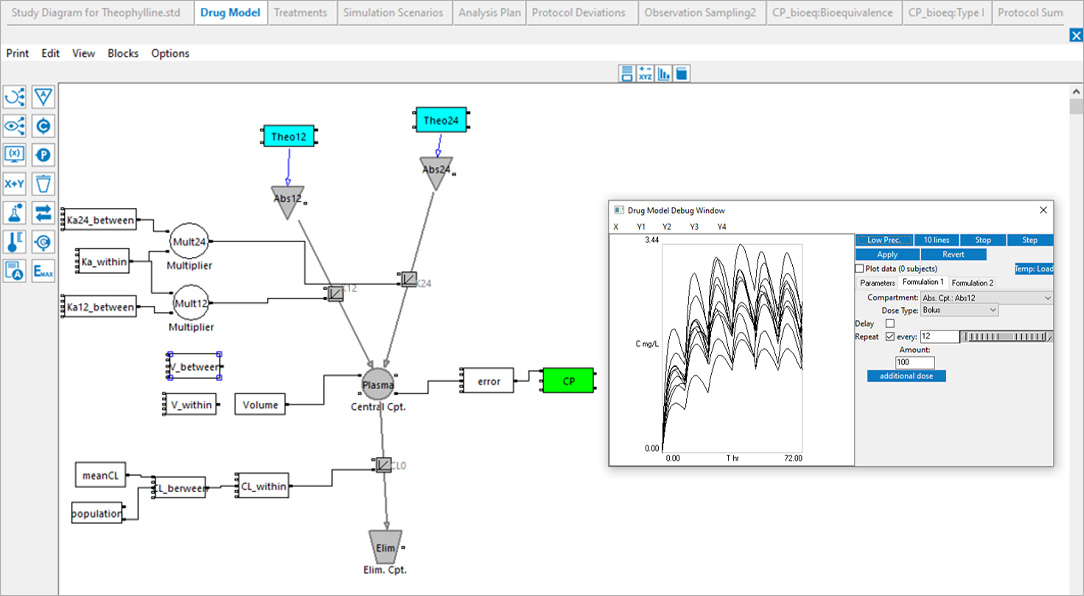
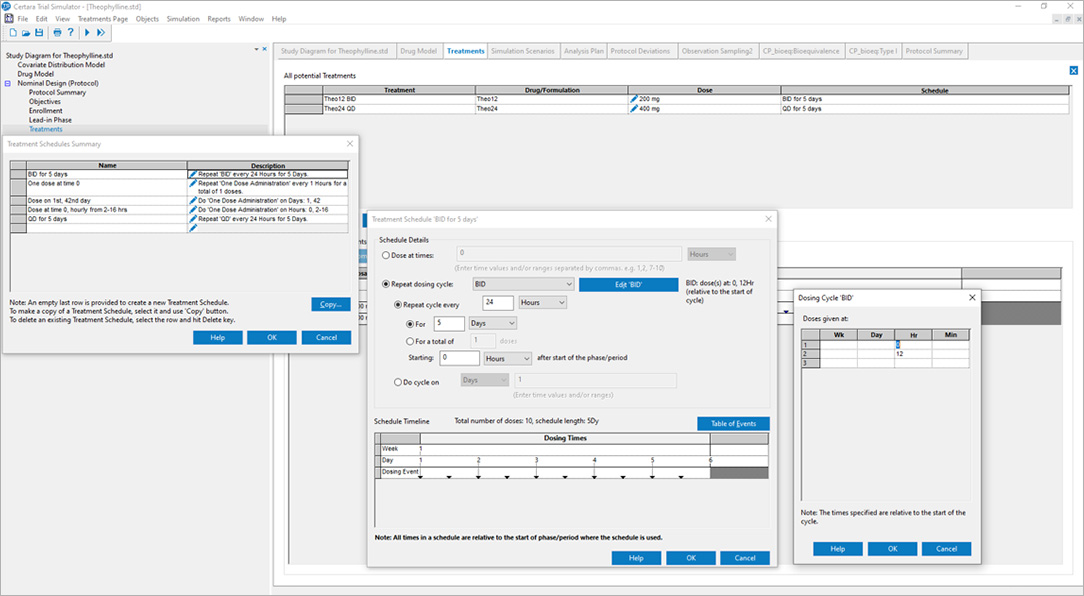
Certara’s Clinical Trial Simulator has been trusted for over a decade by leading pharmaceutical companies to optimize trial design and maximize the probability of success. By leveraging simulation and existing knowledge of the drug under study, Trial Simulator provides critical insights to improve study endpoints and reduce trial costs.