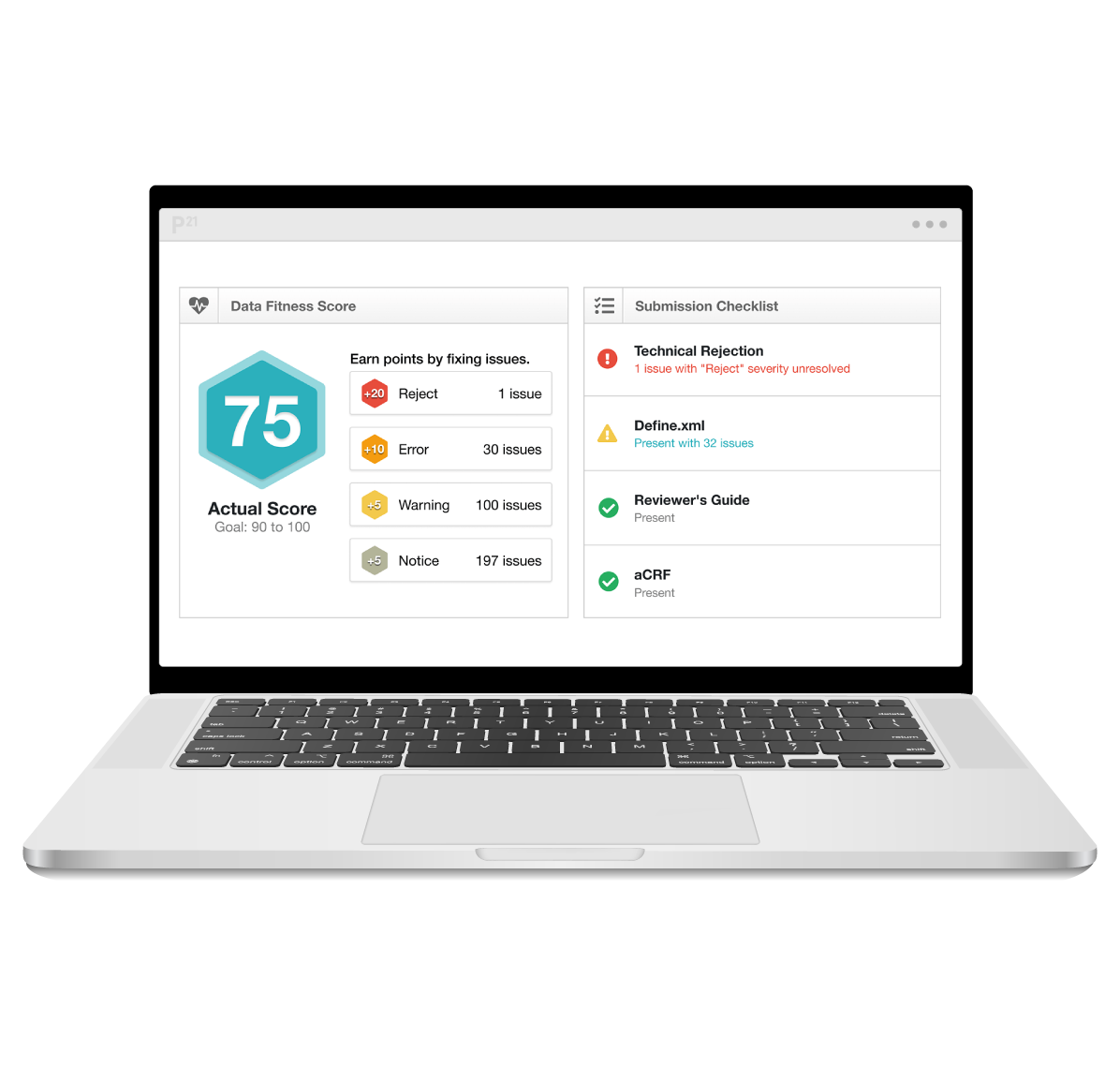
Let our clinical metadata repository transform your clinical data into high-quality, submission-ready deliverables. By combining metadata management and continuous data validation in a collaborative platform, you’ll accelerate timelines, reduce the risk of costly delays, and ensure end-to-end compliance.
Pinnacle 21®
Personalized demo of Pinnacle 21 clinical data standardization software
Driving results for leading pharma organizations




"Pinnacle 21’s MDR is very intuitive and user-friendly, so there is no steep learning curve like you might get with other systems. The integrated validation mechanism means we can do real-time validation, right from the start.”

Radhika Kale
Associate Director of Statistical Programming Standards, Alnylam Pharmaceuticals


