Since modern drug development, drug concentration assays have almost exclusively used plasma as a matrix rather than whole blood. Various theories about assay sensitivity, matrix interference, protein binding, and free drug movement have been put forth to explain why it is “best” to measure drug concentrations in plasma. Personally none of these theories convinces me that plasma drug measurements are superior to blood concentration measurements. However, it is important to understand the difference between blood and plasma measurements, and how they are related. Then you can make an educated decision about which method to use in your drug development situation.
Blood is a fluid that carries nutrients and oxygen to the body and is pumped to its destinations by the heart. Blood is primarily composed of red blood cells (RBC), carriers of nutrients and oxygen, and a liquid called plasma and a few white blood cells, which fight disease. Because white blood cells make up a very small portion of the blood, we will group them with red blood cells for this discussion. The hematocrit is a measure of the proportions of blood that are either cellular (RBC) or liquid (plasma). Specifically, hematocrit is the percentage of blood volume that is cellular, with a minimum value of 0% and a maximum value of 100%. Normal hematocrit is approximately 48% for men and 38% for women. This means that men normally have 48% RBC and 52% plasma. The numbers for women are 38% RBC and 62% plasma.
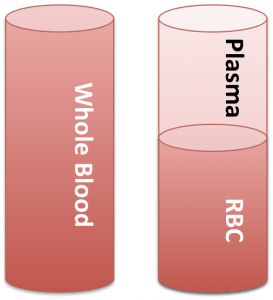
Blood, plasma and red blood cells
When measuring the drug concentration in plasma, a sample of whole blood is drawn from the patient (like cylinder on the left). The sample is then centrifuged to separate the RBC and plasma (like the cylinder on the right). The plasma is withdrawn and analyzed for drug concentration and the RBC is discarded. So, how are blood and plasma concentrations related? Simple, using basic math and some proportionality factors we can convert between these two values.
Equation 1
Equation 2
Now, let’s assume that the drug partitions equally into the RBC and plasma, giving the following relationship:
Equation 3
In this case, the concentration in blood is equivalent to the concentration in plasma. This makes it very easy to “convert” between blood and plasma data. If we now assume that the drug is isolated in the plasma, then we get the following relationship:
Equation 4
In this case, we can substitute in Equation 2 and arrive at the following:
Equation 5
Equation 6
Equation 7
This means that the concentration in blood will be lower than the concentration in plasma. The adjustment factor is 1-Hct. This makes sense because the volume of blood is larger than the volume of plasma, and the amount of drug in both blood and plasma is equivalent. Finally, the last scenario occurs if the drug is sequestered in the red blood cells. To address this, we perform similar manipulations as Equations 5, 6, and 7, giving:
Equation 8
In this situation, blood concentrations are less than RBC concentrations, with Hct as the adjustment factor. This also implies that plasma concentrations will be very small (close to zero) relative to the blood concentrations.
After looking at these equations, what stands out for me is that whole blood concentrations always represent the total amount of drug in the circulatory system. Plasma concentrations are valuable, but only when drug is not sequestered in the red blood cells. In these situations, plasma concentrations are very misleading. One example of a drug like this is brinzolamide (Azopt) used for the treatment of elevated intraocular pressure in glaucoma patients.
In my opinion, whole blood concentrations may provide value for the pharmacokineticist by describing the total drug content in circulatory system, and allowing the ability to convert between plasma and blood when necessary.
The methods used to characterize the pharmacokinetics (PK) and pharmacodynamics (PD) of a compound can be inherently complex and sophisticated. PK/PD analysis is a science that requires a mathematical and statistical background, combined with an understanding of biology, pharmacology, and physiology. PK/PD analysis guides critical decisions in drug development, such as optimizing the dose, frequency and duration of exposure, so getting these decisions right is paramount. Selecting the tools for making such decisions is equally important. Fortunately, PK/PD analysis software has evolved greatly in recent years, allowing users to focus on analysis, as opposed to algorithms and programming languages.


