May 8, 2026

Jen Manzi
Subject Matter Expert and User Advocate, Pinnacle 21 by CertaraJen Manzi is a Subject Matter Expert and User Advocate at Pinnacle 21. She has over 20 years of Pharma/Life Sciences industry experience in Clinical Trials and Safety Data Management. Jen has held various roles within these areas, including eCRF Programmer, SDTM Delivery Lead, Product Owner and Programmer of Batch Processes, Vendor Relationship Manager, Program and Process Improvement Manager, and Validation Lead.
Deliver compliance assurance for every sponsor
Learn more about how CROs can effectively manage submission complexity at scale with Pinnacle 21 Enterprise.

Make an inquiry about Pinnacle 21 Enterprise
Pinnacle 21 Enterprise builds on Community’s core validation capabilities with:
Make an inquiry to learn how your organization can benefit from reduced risk, increased quality, and guided submission readiness with Pinnacle 21 Enterprise.
FAQs
Why are Phase II and III trials more complex than Phase I?
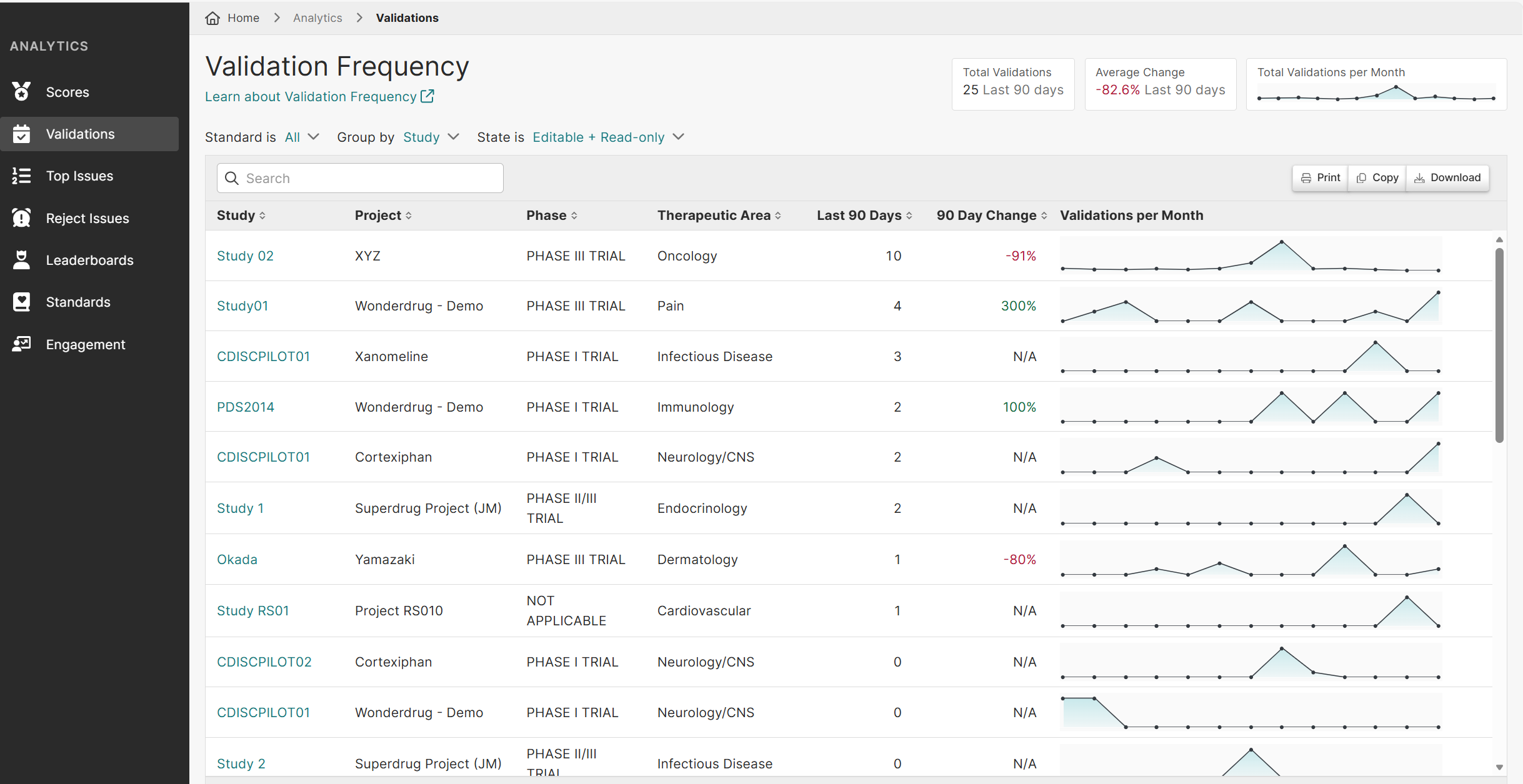
Phase II and III trials are more complex because they involve larger patient populations, more endpoints, and multiple sites. For a late phase CRO, this significantly increases data volume and introduces more cross-domain dependencies, making late phase clinical data validation and overall late phase clinical data management far more demanding.
What is CRO data management and why is it critical in late-phase trials?
CRO data management refers to the processes and systems used by contract research organizations to collect, validate, standardize, and prepare clinical trial data for analysis and regulatory submission. In late-phase trials, effective CRO regulatory compliance tools are essential due to the scale and complexity of the data involved. They ultimately enable high-quality, compliant datasets that support reliable results and timely submissions.
What is the biggest data management challenge for CROs in late-phase trials?
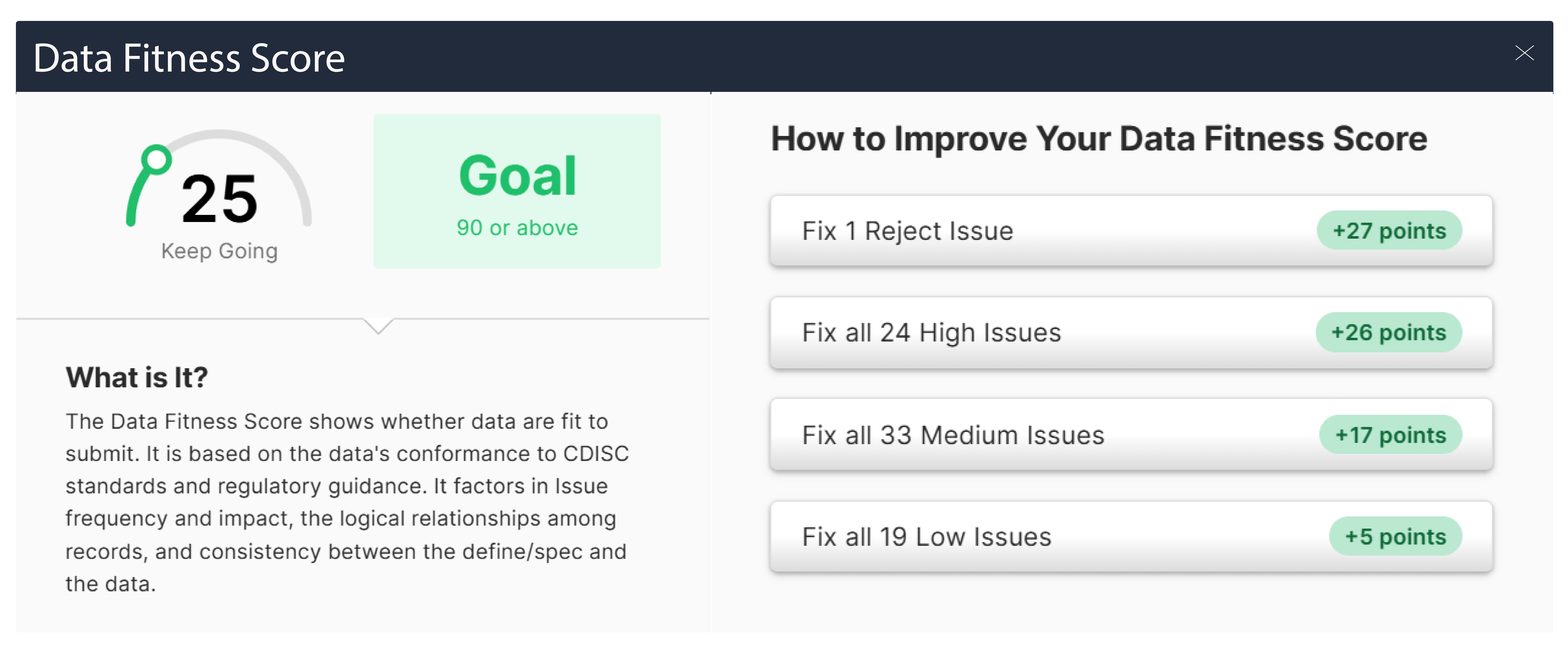
For a late phase CRO, the biggest challenge is managing large, complex datasets while maintaining consistency, quality, and compliance under tight timelines. Effective late phase clinical data validation is critical, as increasing data volume and validation rules can quickly lead to inefficiencies and delays.
How does Pinnacle 21 Enterprise improve submission readiness?
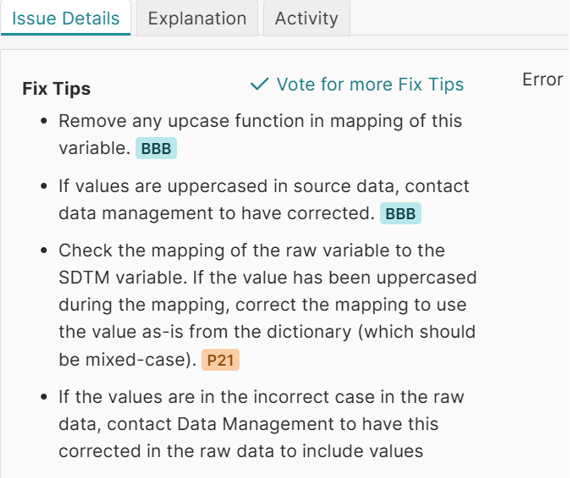
Pinnacle 21 Enterprise supports late phase clinical data validation by enabling continuous validation, early issue detection, and standardized workflows. For a late phase CRO, this improves data quality and reduces last-minute delays, while supporting stronger regulatory alignment through advanced CRO regulatory compliance tools. Learn more about how Pinnacle 21 Enterprise supports submission readiness for CROs in this fact sheet: https://www.certara.com/fact-sheet/how-to-improve-cro-data-management/
Can Pinnacle 21 Enterprise support multiple sponsors?
Yes. Pinnacle 21 Enterprise enables a late phase CRO to manage multiple studies and sponsors simultaneously with consistent rule application and scalable processes. This ensures efficient late phase clinical data validation across programs while maintaining high standards of quality and compliance. Learn how CROs can scale efficiently while maximizing validation assurance in this blog: https://www.certara.com/blog/platforms-for-collaboration-between-sponsors-and-cros-5-ways-cros-can-scale-efficiently-while-maximizing-validation-assurance/
You May Also Like
This is custom heading element
This is custom heading element
Lorem ipsum dolor sit amet consectetur. Donec diam risus volutpat tempor. Massa accumsan aliquam nibh tortor.Lorem ipsum dolor sit amet consectetur. Donec diam risus volutpat tempor.
This is custom heading element
Lorem ipsum dolor sit amet consectetur. Donec diam risus volutpat tempor. Massa accumsan aliquam nibh tortor.Lorem ipsum dolor sit amet consectetur. Donec diam risus volutpat tempor.
This is custom heading element
Lorem ipsum dolor sit amet consectetur. Donec diam risus volutpat tempor. Massa accumsan aliquam nibh tortor.Lorem ipsum dolor sit amet consectetur. Donec diam risus volutpat tempor.





