March 25, 2026
The limitations of static Global Value Dossiers in pharma
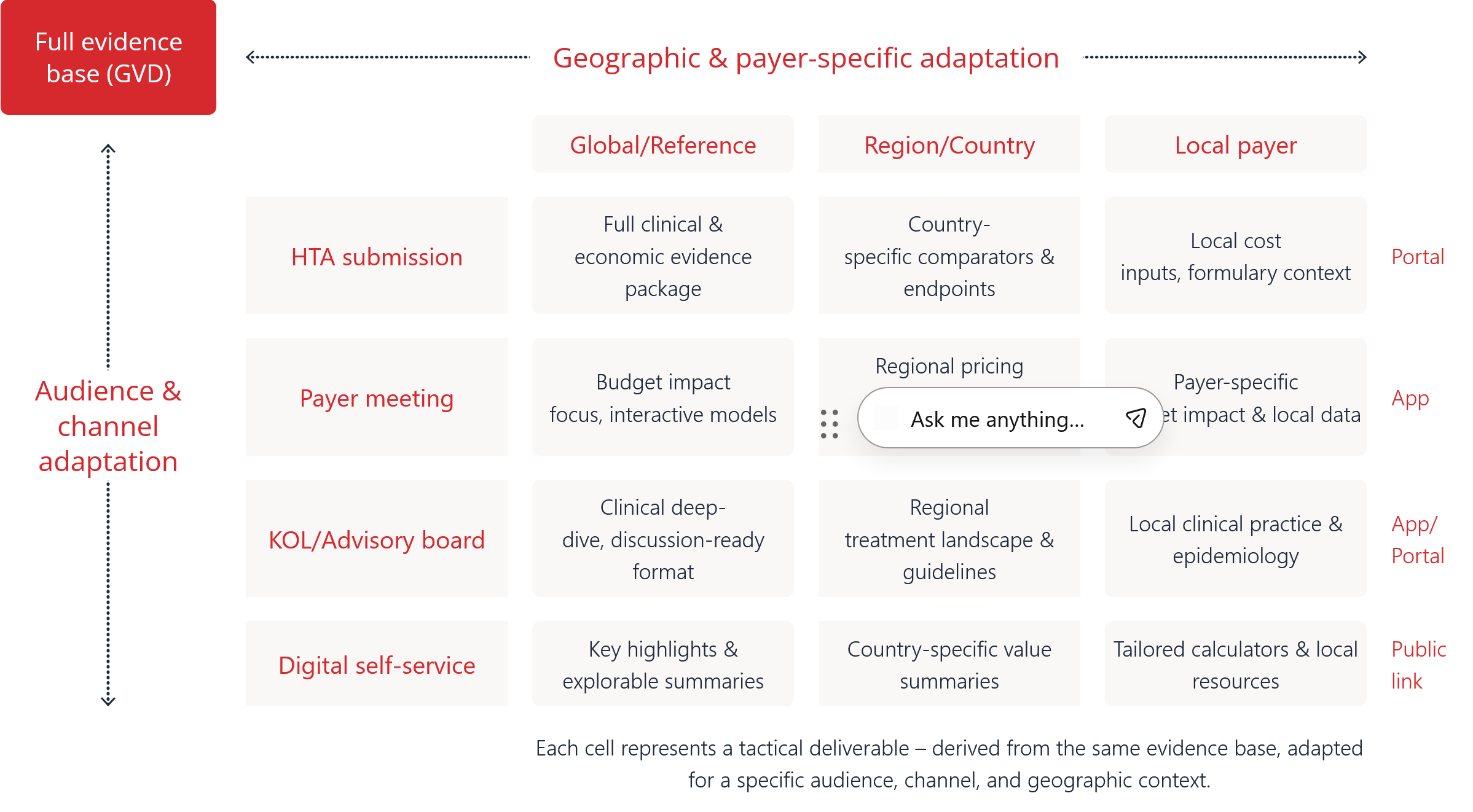
A payer evaluating budget impact is looking for different things than an HTA reviewer focused on clinical endpoints or a Key Opinion Leader (KOL) preparing an advisory board. A static document offers one path through the content, regardless of who’s reading it.
Field teams still present in person, but more frequently share materials remotely. Stakeholders increasingly review evidence ad hoc, not in parallel, using whatever device is at hand. A single static format doesn’t effectively serve all of these workflows.
Clinical, economic, and humanistic data are interconnected. A PDF enforces a sequential reading experience. It’s hard for stakeholders to navigate directly to what matters to them, explore supporting data, or interact with the assumptions behind an economic model.

Prove value faster with interactive evidence
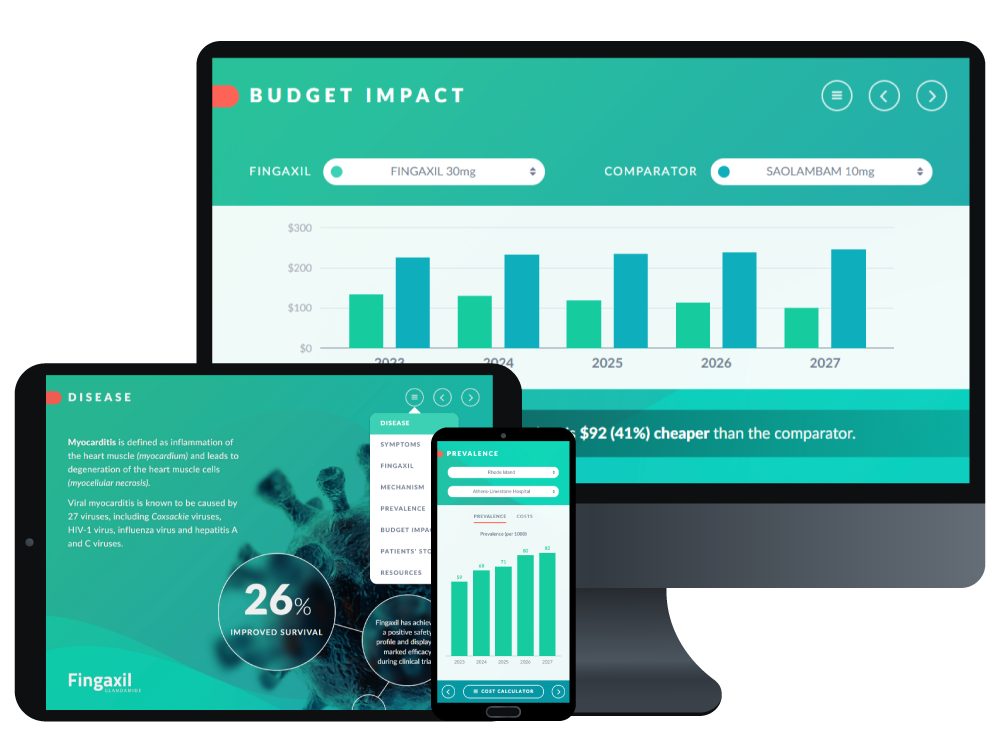
See how BaseCase iGVD software helps teams streamline evidence communication, reduce prep time, and engage payers more effectively.
When both axes are leveraged strategically, the result is a suite of tactical materials derived from a single source of truth but tailored for its specific audience, channel, and geography.

Ready to prove value faster?
Explore how an interactive global value dossier app transforms complex evidence into a structured, engaging experience. You’ll see how BaseCase iGVD software helps teams align on the latest content, reduce prep time, and communicate value more quickly to payers and decision makers.
This blog was originally published on January 24, 2024 and has been updated for accuracy.

Christian Pichardo
Director, Product ManagementChristian Pichardo serves as Director of Product Management at Certara/BaseCase. He leads the design and strategic direction of digital solutions that strengthen health economics and value communication, ensuring alignment with customer needs and market trends.
Before stepping into his current role, Mr. Pichardo spent more than 15 years working across the life sciences, consulting, and software industries, holding leadership roles in market access, health economics, and product strategy. In addition to his corporate experience, he has co-founded consulting ventures focused on healthcare innovation and capability development, further broadening his perspective on advancing access and value in healthcare.
Mr. Pichardo holds degrees in Economics and Applied Mathematics from the Instituto Tecnológico Autónomo de México (ITAM) and a Professional Certificate in Product Management from the Massachusetts Institute of Technology (MIT). He has published peer-reviewed research on cost-effectiveness and real-world outcomes in leading scientific journals.
Make an inquiry
Ready to bring your value story to life? Make an inquiry to learn how BaseCase value communication software transforms complex economic models and clinical data into interactive, field-ready tools.
FAQs
What is an interactive Global Value Dossier and how does it improve market access?
An interactive Global Value Dossier is a digital, user-friendly version of a traditional GVD that allows stakeholders to easily navigate and customize content. This kind of value dossier automation improves market access by allowing key stakeholders to:
- Quickly access relevant evidence
- Adapt value messages in real time
- Ensure consistent communication across markets
What are the key steps in value dossier preparation?
Global value dossier preparation involves both defining the value strategy and compiling the evidence needed to support it. This includes gathering clinical efficacy and safety data, health economic models, burden of disease insights, and comparator analyses, alongside developing clear value messages for payers. The process also covers cross-functional alignment, quality review, and structuring content for easy localization across different healthcare systems. A well-structured preparation process ensures consistency, efficiency, and readiness for payer engagement.
When should a Global Value Dossier be developed during drug development?
Health value dossiers are typically initiated during late-phase clinical development (Phase II/III) and refined as more data becomes available. Early development ensures alignment between evidence generation and payer expectations, while ongoing updates keep the dossier relevant for launch and post-launch market access activities.
How is a Global Value Dossier used in local market access submissions?
Local affiliates can adapt the GVD to meet country-specific requirements, such as HTA submissions or reimbursement dossiers. While the global narrative remains consistent, elements like comparator data, epidemiology, and cost inputs can be tailored. This ensures alignment with local payer expectations while maintaining a unified global value story.
How does value dossier automation enhance efficiency and consistency?
Value dossier automation uses digital platforms and structured content systems to streamline updates, version control, and localization. It reduces manual effort, minimizes errors, and ensures consistent messaging across markets. Automation also helps teams maintain up-to-date and compliant dossiers throughout the product lifecycle.
Why work with trusted health economics partners for value dossiers?
Trusted health economics partners bring specialized expertise in evidence collation, economic modeling, and payer expectations. Collaborating with experienced interactive global value dossier platform providers helps ensure that value dossiers are robust, aligned with HTA requirements, and tailored to relevant markets. This significantly improves the quality and credibility of health value dossiers, supporting more successful reimbursement outcomes.