Many factors make it challenging to obtain adequate pediatric samples for use in drug development and to correctly determine doses. Age is an important factor because a drug may act very differently depending on the stage of the child’s development.
Pharmacokinetic (PK) and pharmacodynamic (PD) studies are employed to determine optimal drug dosing in children and understand how the body affects the drug and how the drug affects the body. Children could have different PK and PD profiles compared to adults, which necessitates age-appropriate dosing.
In addition, many parents may not consent to have their children enrolled in clinical studies. Thus, there are often inadequate sample sizes when it comes to assessing the PK for biologics, drug-drug interactions (DDIs), and dose determination.
It is not as simple as giving a slightly smaller drug dose to a child. This is where mathematical modeling can provide valuable information.
Physiologically-based pharmacokinetic (PBPK) models are mathematical models that simulate the effect of the human body on drugs; at Certara, we use the Simcyp PBPK Simulator. These models account for various factors, including absorption of the drug, its distribution within the body, and its elimination. PBPK models are used to predict the concentration of a drug in various tissues and organs and to understand how a drug is metabolized. In addition, they can be used to evaluate a new drug’s safety or determine the best dose.1
Predicting Drug Exposure During Breastfeeding
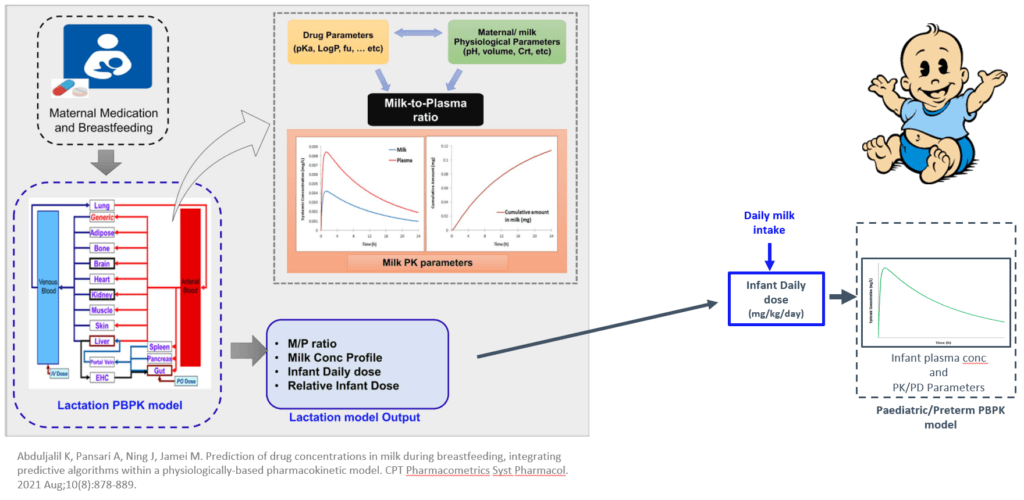
There has been increasing interest in using PBPK models to determine the safety and efficacy of drugs in breastfeeding mothers and infants. Pregnant and breastfeeding patients are generally advised to avoid pharmacological treatments. However, this isn’t always possible because stopping a medication can have adverse effects on some medical conditions. Therefore, predicting what will happen to a fetus or newborn when exposed to medication is crucial.2, 3
Lactation PBPK models can help predict the concentration of a drug in breast milk, which is then used to estimate an infant daily dose consumed during breastfeeding. This information can be used to help determine the exposure of a drug in breastfeeding infants and help lactating women make informed decisions about whether to breastfeed.4
PBPK Modeling in Pediatrics
PBPK models are also widely used in pediatric drug development and dose selection. They are also becoming more widely accepted by regulatory agencies to evaluate new pediatric drug candidates by evaluating both toxicity and efficacy and predicting the extent of exposure.6
PBPK modeling in pediatrics facilitate investigating the effects of age-related changes on pharmacokinetics and drug disposition.They allow us to ask and answer many questions regarding what factors are important when examining exposure in neonates, infants, and older children.
This approach can be used to eliminate the need for off-label prescribing when prescribing information is not available. To learn more about PBPK modeling in drug development for children and pregnant women, please register for this upcoming LinkedIn Live event.
References:
- Wang K, Jiang K, et al. Physiologically based pharmacokinetic models are effective support for pediatric drug development. AAPS PharmSciTech. 2021;22(6). Accessed August 23, 2022.
- Coppola P, Kerwash E, Cole S. Physiologically based pharmacokinetics model in pregnancy: A regulatory perspective on model evaluation. Frontiers in Pediatrics. 2021;9. Accessed August 22, 2022.
- Coppola P, Kerwash E. Physiologically based pharmacokinetics model in pregnancy: A regulatory perspective on model evaluation. Frontiers in Pediatrics. 2021;9.
- Abduljalil K, Pansari A, et al. Prediction of drug concentrations in milk during breastfeeding, integrating predictive algorithms within a physiologically-based pharmacokinetic model. CPT: Pharmacometrics & Systems Pharmacology. 2021;10(8):878-889. Accessed August 23, 2022
- Stamatopoulos K, Pathak S,et al. Population-based PBPK model for the prediction of time-variant bile salt disposition within gi luminal fluids. Molecular Pharmaceutics. 2020;17(4):1310-1323. Accessed August 22, 2022.
- Johnson TN, Small BG, Rowland Yeo K. Increasing application of pediatric physiologically based pharmacokinetic models across academic and industry organizations. CPT Pharmacometrics Syst Pharmacol. 2022;11(3):373-383. doi:10.1002/psp4.12764


