Would your drug development program benefit from partnering with experts in scientific and medical communications and publications? Maybe, we should first address what exactly scientific and medical communications and publishing entail.
As medical writers with decades of experience, we can say that every day holds unique, intellectually rigorous challenges. Our work includes the following:
- Publication planning;
- Providing advice on scientific journal selection and congress participation;
- Writing abstracts, posters, and manuscripts;
- Coordinating and incorporating author feedback on draft documents;
- Submitting manuscripts to journals and abstracts and posters to congresses; and
- Developing responses to journal reviewer comments on submitted manuscripts.
Sometimes, we do all these things in a single day!
Medical Publishing
Clinical and nonclinical researchers publish their work in peer-reviewed journals and as posters and presentations at congresses. These are our forums for communicating research findings to other scientists, healthcare professionals, and patients. Sharing evidence that demonstrates that a new therapy helps patients is exciting!
New treatments or treatments with improved safety/efficacy can’t help patients until prescribers learn your results. And that is what a medical publications professional does every day.
Scientific Communication
Communicating new data can be a daunting experience, and it should be! The transparent, accurate, and timely communication of medical information is key to drug development.
Industry guidelines specify what to report, when to report, and authorship, among other things. The guidelines help ensure that research findings can be independently confirmed. Good Publication Practices (GPP3), ICMJE recommendations, and CONSORT are some of the published guidelines that medical publication professionals follow when developing projects.
Some days, we are helping to update the publication guidelines. For example, experienced medical publication professionals provided input for the GPP3 update.
Figure 1: From Concept to Publication
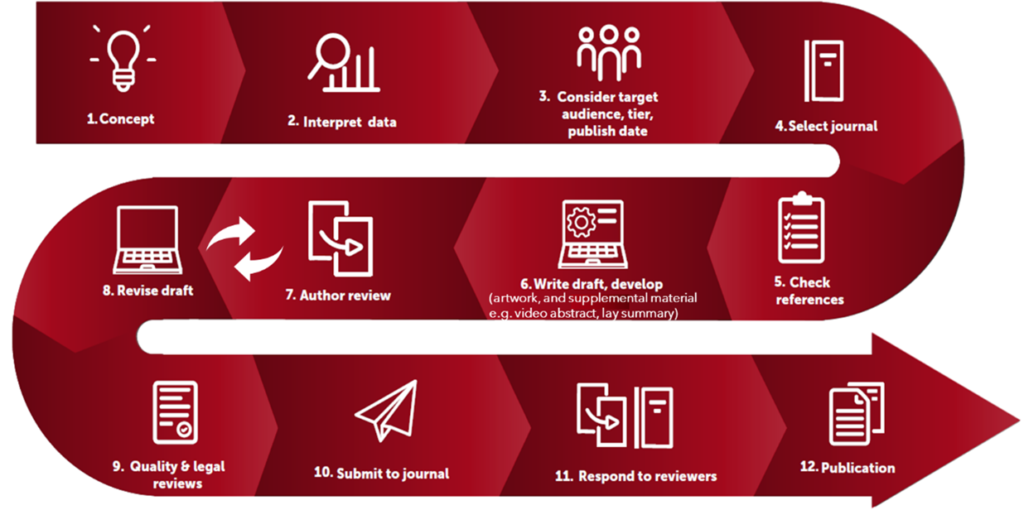
Publications Process
Adherence to industry guidelines and the publications process (Figure 1) helps to ensure the development of quality publications. From concept to publication, a medical publications professional supports you throughout the process. With our wealth of experience with clinical research in different therapeutic areas, with different types of journals, and with diverse authoring teams, we streamline the process. On any given day, a writer might support 1 or all 12 steps from concept to publication.
Publication Planning
Medical publication professionals also help you get to the concept stage of publication writing (i.e., tactical execution) by working with you on publication planning. Publication professionals think about and do publication planning every day including:
- Developing key messages for a clinical development program
- Determining the timing of data disclosures to congresses and development of related manuscripts
- Selecting a journal based on the desired audience, time to publication, and journal impact
- Matching the right writer to a given project
- Developing a plan for review of disclosures within a drug development company.
Planning is the most important part of our job. Thinking ahead, anticipating the next steps, and adhering to timelines underpins everything we do. This planning is critical to the successful execution of a publication plan.
The Certara Team
We have a great team of medical publication professionals who are also highly skilled at project and time management. Many of us are also Document Quality Checkers. So sometimes our workday involves reading, reviewing, and editing documents developed by other writers. We review our documents’ grammar, spelling, and accuracy so that our clients can have confidence in their quality.
We also regularly share knowledge, learnings, and best practices with each other on the Certara Medical Publications team. Each member of our team of professionals brings a unique perspective and passion for medical publications writing.
We come to work every day eager to learn new things and support the publication of clinical data to enhance patient care. Hopefully, this description of a day in the life of a medical publications professional has illustrated how the Certara team can support your program’s scientific communication needs.
For an example of how our team collaborated with our Certara clinical pharmacology colleagues to support a drug development program for migraine, read this case study.
This blog was originally published on June 1, 2022. It was updated on July 19, 2024.




