Tag
Evidence and Access
CMS Cell and Gene Therapy (CGT) Access Model Explained: The Most Significant Drug Pricing Legislation You’ve Never Heard Of

December 13, 2024 The Centers for Medicare & Medicaid Services (CMS) is piloting a program…
CertaraDecember 13, 2024
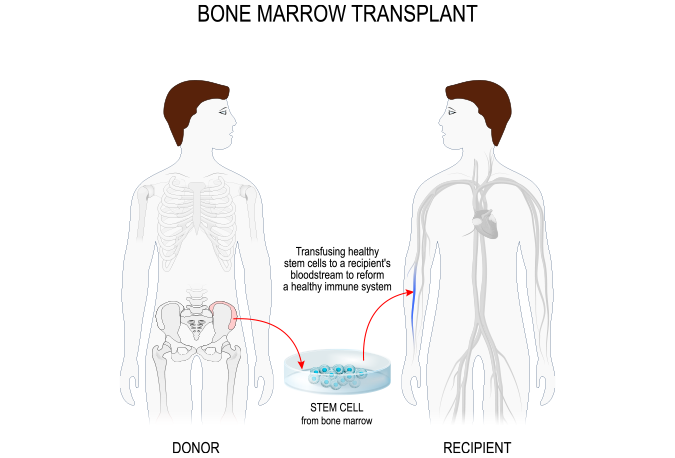
Joint Clinical Assessment (JCA) And Potential Consequences For The Evaluation Of Gene Therapies

In October 2019, Galderma announced that the U.S. FDA approved AKLIEF® (trifarotene) Cream, 0.005% for the…
CertaraJune 17, 2024
Accelerating a drug launch with value communication technology and an integrated workflow

With early approvals for a targeted cancer drug in Brazil and Argentina, this global pharmaceutical…
CertaraOctober 2, 2023
ICER’s Unsupported Price Increase Report: Unfit for Policymaking

Since 2019, the Institute for Clinical and Economic Review (ICER) has published an annual Unsupported…
CertaraJuly 25, 2023
Using Real-World Evidence to Derive an External Control Arm for a Rare Disease Drug Trial

Our client was developing a new anti-viral drug for a rare, life-threatening viral infection that…
CertaraApril 17, 2023
Using Real Word Evidence to Enhance Drug Development

Real-world evidence (RWE) has emerged as an increasingly important tool in the drug development process,1…
CertaraApril 5, 2023
Agent-Based Model Predicts Impact of Monoclonal Antibody Treatment, Prophylaxis & Vaccines on Viral Transmission during Pandemic

Agent-based models focus on the heterogeneous behavior of individual agents and their interactions with one…
CertaraMarch 2, 2023
Defining path-to-market strategy through a data-driven and scientific approach

How Certara supported a gene therapy manufacturer by using a selection of gateway and value…
CertaraOctober 28, 2021
The Scary Future of Rare Disease Management

In a world of rising healthcare costs, the greatest nightmare for payers is reimbursing drug…
CertaraSeptember 23, 2021