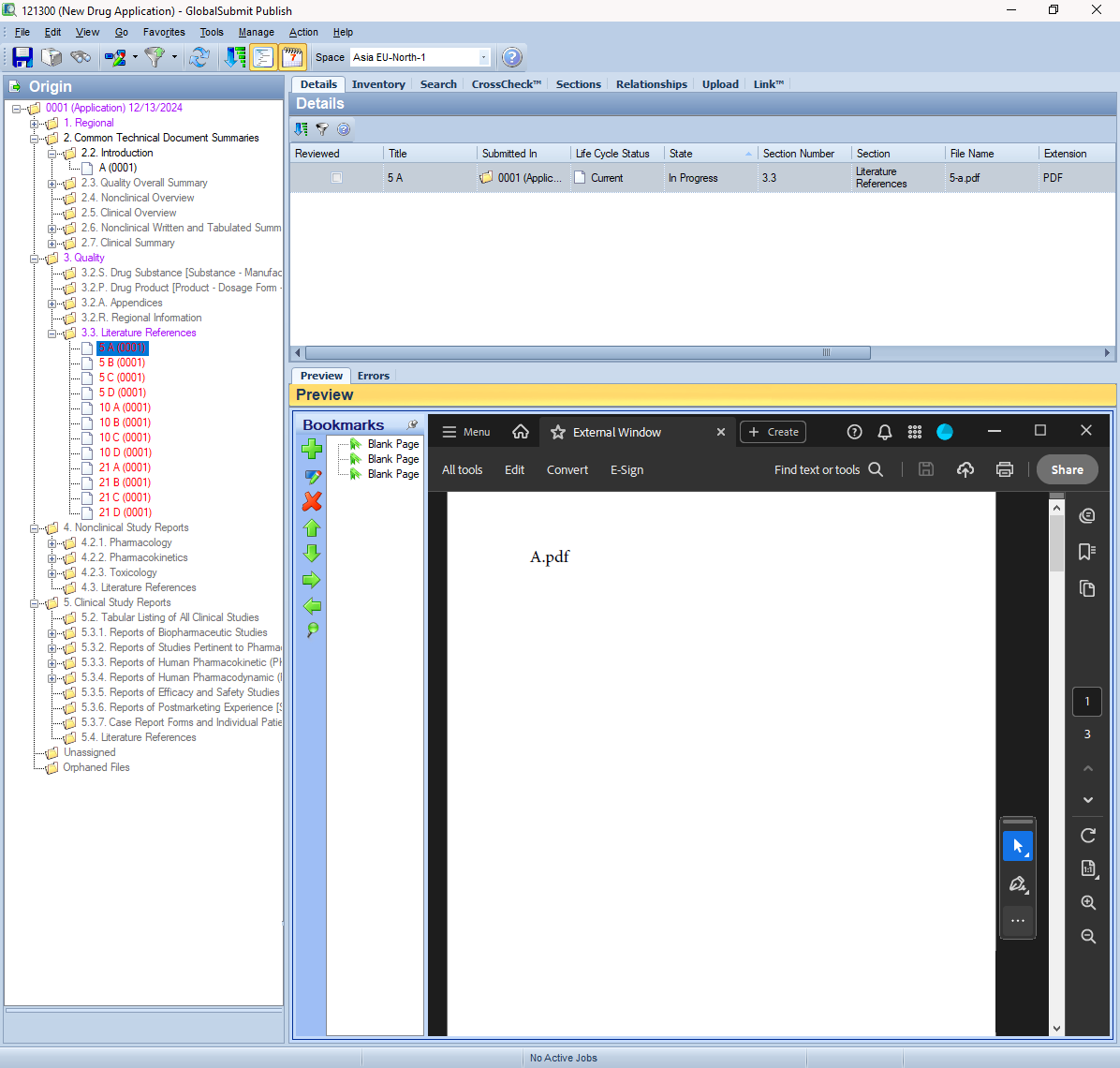
GlobalSubmit PUBLISH is a cutting-edge eCTD publishing software that empowers regulatory teams to create and submit compliant eCTD submissions efficiently to health authorities worldwide. Designed with advanced features like live validation, automated hyperlink creation, and accelerated quality control, this software builds quality and efficiency into the eCTD submission process. Whether you’re an experienced publisher or new to regulatory submissions, GlobalSubmit PUBLISH ensures accuracy, compliance, and significant time savings.
GlobalSubmit™ PUBLISH
Generate compliant submissions with our advanced eCTD publishing software
Learn more about GlobalSubmit PUBLISH
Quality at speed
Efficiency in eCTD publishing is critical as your regulatory department races to meet deadlines and deliver treatments to patients. GlobalSubmit PUBLISH simplifies this process by reducing manual tasks, ensuring compliance, and accelerating workflows. With features like Live Validation, automated PDF processing, and side by side confirmation of bookmark and hyperlink destinations for quality control, this software is a game-changer for regulatory professionals.
Simple, efficient, and transparent
Real-time validation
Live Validation checks for over 200 error conditions, including 40+ PDF checks, ensuring your submissions meet technical requirements and avoid rejection.
Automated PDF processing
Eliminate manual publishing tasks with auto-processed, regulatory-compliant PDFs, saving time and ensuring accuracy.
Accelerated QC process
Reduce QC time by 8X and improve accuracy by 3X, enabling faster and more reliable submissions.
PUBLISH in the latest standard
View our webinar series on eCTD 4.0 to understand the new standard and learn how GlobalSubmit is your key to adopting it.

Related resources
View all
With GlobalSubmit PUBLISH, your data is secure
Certara holds ISO 27001 certification for Certara’s Information Security Management System (ISMS). We have implemented robust security controls, undergone rigorous risk assessments, and continuously strive for improvement. GlobalSubmit PUBLISH ensures full compliance with global data protection standards, offering peace of mind for sensitive analysis.
Schedule a demo
Discover how GlobalSubmit PUBLISH can streamline your eCTD submission process.
FAQs
What is eCTD publishing software?
eCTD publishing software simplifies the creation and submission of electronic Common Technical Documents (eCTDs) to regulatory agencies.
Which health authorities are supported by GlobalSubmit PUBLISH?
GlobalSubmit PUBLISH supports submissions to the FDA, EMA, Health Canada, Swissmedic, TGA (Australia), PMDA (Japan), China, South Africa, and Gulf Coast Council (GCC).
How does live validation improve submission accuracy?
Live validation identifies and corrects errors in real-time, reducing the risk of technical rejection.
What types of documents does GlobalSubmit PUBLISH Support?
GlobalSubmit PUBLISH supports IND, NDA, ANDA, sNDA, BLA, MAA, DMF, ASMF, NDS, and more.


