
Olanzapine is an effective antipsychotic medication used to treat schizophrenia, a disease that affects about 1 in 300 people worldwide.1 However, side effects such as weight gain can cause patients to become less likely to take the medication as directed.2
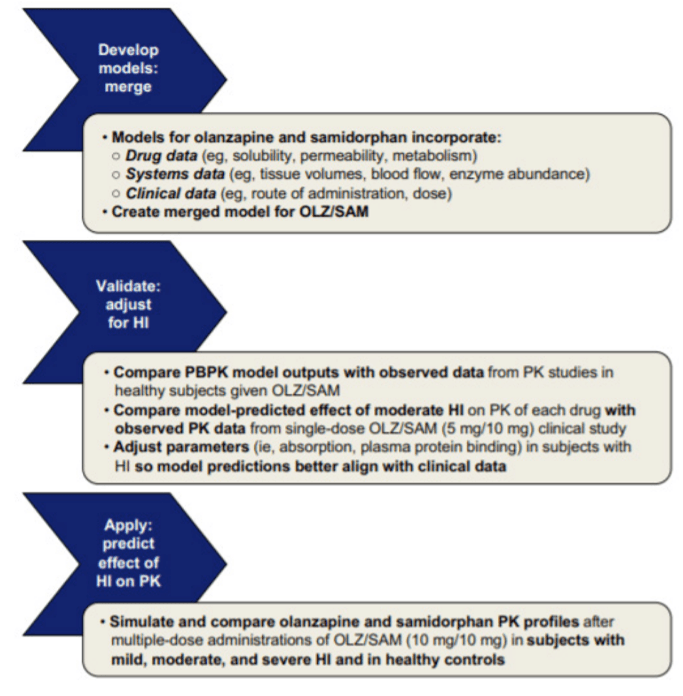
Modeling can predict the role that hepatic function has on olanzapine/ samidorphan
Hepatic metabolism plays a major role in both olanzapine and samidorphan clearance. Impaired hepatic function could affect the PK of both compounds. To assess this risk, the Simcyp PBPK model was refined to predict changes in olanzapine and samidorphan PK after multiple once-daily doses of OLZ/SAM in subjects with mild, moderate, and severe hepatic impairment.5
To evaluate the PK changes in subjects with moderate hepatic impairment, model parameters such as absorption rate constant and fraction unbound to plasma protein were modified. PBPK model outputs were compared with observed data from PK studies in healthy subjects given OLZ/ SAM.
The PBPK modeling indicated that mild hepatic impairment would have minimal impact on steady-state exposures of olanzapine and samidorphan, and moderate to severe hepatic impairment would result in up to 1.6-fold and 2.3-fold increases in total exposure (area under the curve; AUC) of olanzapine and samidorphan, respectively. PBPK modeling allowed for predicting untested clinical scenarios of varying degrees of hepatic impairment in lieu of conducting additional clinical studies.
- World Health Organization. Schizophrenia.
https://www.who.int/news-room/fact-sheets/detail/schizophrenia
Published 2022. Accessed September 10, 2022. - Komossa K, Rummel-Kluge C, Hunger H, Schmid F, Schwarz S, Duggan L, Kissling W, Leucht S. Olanzapine versus other atypical antipsychotics for schizophrenia. Cochrane Database Syst Rev. 2010 Mar 17;(3). Accessed on September 10, 2022.
- Certara. Simcyp Discovery Simulator. 2022. Accessed 10 September, 2022. https://www.certara.com/app/uploads/2022/07/BR-Simcyp-Discovery.pdf.
- Lei S, L, von Moltke, L, Rowland Yeo, K, Physiologically-Based Pharmacokinetic Modeling for Predicting Drug Interactions of a Combination of Olanzapine and Samidorphan, CPT Pharmacometrics Syst Pharmacol. 2020 Feb;9(2):106-114. Accessed on September 10, 2022.
- Sun L, Barter Z, von Moltke L, Rowland Yeo K. Using physiologically-based pharmacokinetic modeling for predicting the effects of hepatic impairment on the pharmacokinetics of olanzapine and samidorphan given as a combination tablet. CPT Pharmacometrics Syst Pharmacol. 2021 Sep;10(9):1071-1080. Accessed on September 10, 2022.
Contact us




