Within drug applications (ANDA, BLA, IND, or NDA), eCTD (electronic common technical document) Module 3 pertains to quality data. It has become increasingly common for sponsors to reference a Drug Master File (DMF) within this module. A DMF contains detailed information about facilities, processes, or articles used in the manufacturing, processing, packaging, and storing of drugs or biologics. This allows the manufacturer of a component of the drug or biologic to share proprietary information with the US FDA without having to share the information with the sponsor directly.
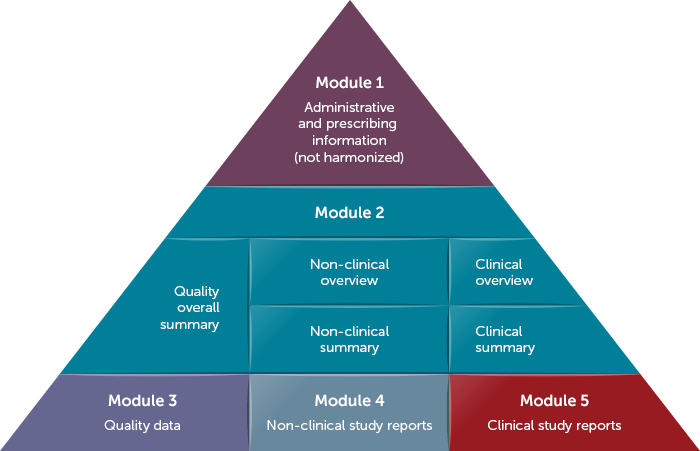
Modular Structure of Common Technical Document
However, referencing a DMF in a regulatory application requires careful coordination between the DMF owner and the application’s sponsor. In this blog post, we will discuss some best practices for placing the DMF reference letters in an application.
When a DMF reference occurs, both the DMF owner and the application’s sponsor must appropriately inform the FDA.
In “The Comprehensive Table of Contents Headings and Hierarchy” for US eCTD Module 1, we have these two relevant sections:
- 1.4 references
- 1.4.1 Letter of authorization
- 1.4.2 Statement of right of reference.
Many people find it difficult to distinguish between these sections and decide where to place their LOA. Our experience is that sponsors often get this wrong. But you can always choose the correct section by thinking about what the action is that you are doing, and why you are informing the FDA. Are you giving (authorizing) or receiving (referring to) information?
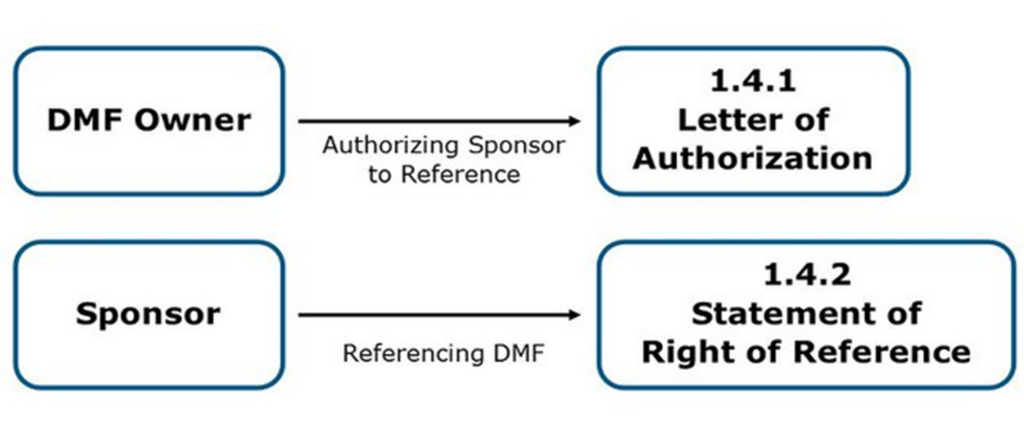
- A DMF owner grants an application’s sponsor the right to reference the DMF by sending them a Letter of Authorization (LOA). The DMF owner must inform the FDA of this by submitting a copy of the LOA in the DMF in eCTD section 1.4.1 Letter of Authorization.
- A sponsor of an ANDA, BLA, IND, or NDA who wishes to incorporate information from a DMF in their application must have received a LOA from the DMF holder. They must submit a copy of the LOA to the FDA in their application in eCTD section 1.4.2 Statement of Right of Reference.
Sponsors who are referencing another application they own (not a DMF) should also use this convention. Simply place the LOA in section 1.4.1 of the application that is to be referenced. Then, place a copy of the LOA in section 1.4.2 of the application that is making the reference.
Follow these best practices for the placement of DMF reference letters to keep your DMFs and applications well organized. Your intentions will be abundantly clear to the FDA.
If you have any questions or need assistance with your submission process, please don’t hesitate to contact our regulatory operations team of experts. We can help you navigate the complex and evolving regulatory landscape, ensure compliance with the latest guidelines, and optimize your submission strategy for faster and smoother approvals. Learn more about our services and how we can support your drug development goals here.
*This blog was originally published by Evan Richardson on March 14, 2016, and was updated on Jan 8, 2023, by Janet Shoshitaishvili.



