Demonstrating bioequivalence (BE) remains the key regulatory hurdle for generic drug approval. However, this process is challenging for complex drugs and alternative delivery methods. This problem is especially vexing for topical drugs and transdermal patches.
The high cost of running clinical BE trials is a major challenge in developing generic versions of topical drugs. As a result, many branded topicals remain on the market well past the originator’s patent expiration date, without the option of cost-effective generic alternatives that could benefit patients. The FDA has noted the problem and has provided new regulatory pathways and incentives such as the Complex Generics Therapy (CGT) to spur development.
Alongside financial incentives, the agency is now turning to in silico modeling & simulation, specifically mechanistic physiologically based pharmacokinetics (PBPK) that leverages in vitro data rather than running an in vivo comparative clinical BE endpoint study.
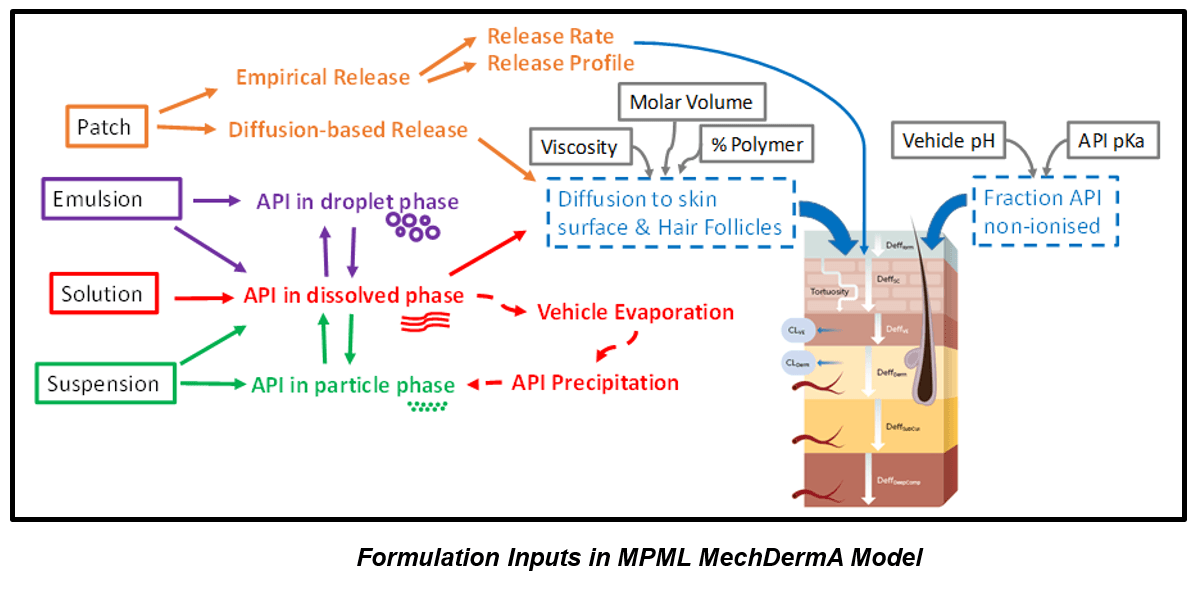
Watch this webinar to learn how Certara expanded its PBPK Simcyp Simulator to incorporate an extensive dermal model— MPML MechDermA. This multi-phase, multi-dimensional dermal absorption model is based on detailed description of skin physiology. The model can simulate drug partitioning and absorption through the hair follicular pathway with blood flow to the dermis modelled as a function of cardiac output, body weight, and body surface area. It also accounts for drug formulation components specific to solutions, emulsions, patches, suspensions, and pastes. Further expansion of the model focuses on a more mechanistic description of the skin to allow simulating disease-specific skin modifications. The model’s formulation module is also continuously enhanced with additional formulation specific critical quality attributes.
VBE leverages advanced modeling and simulation to demonstrate BE and provide additional insight into drug performance. These in silico studies are safer, faster, and less expensive to conduct than clinical BE studies and represent an important advance for generic and innovator drug companies alike.
About Our Speaker
Dr. Sebastian Polak is a Senior Principal Scientist at Certara where he is a member of the modeling and simulation group. He leads the development of cardiac safety modeling and simulation  systems based on the biophysical models of human cardiac myocytes operating at the population level (CSS – Cardiac Safety Simulator). Dr. Polak is also a tenured Associate Professor in Biopharmaceutics at the Faculty of Pharmacy at Jagiellonian University in Krakow, Poland.
systems based on the biophysical models of human cardiac myocytes operating at the population level (CSS – Cardiac Safety Simulator). Dr. Polak is also a tenured Associate Professor in Biopharmaceutics at the Faculty of Pharmacy at Jagiellonian University in Krakow, Poland.


