Nonalcoholic fatty liver disease (NAFLD) is a condition wherein the liver stores excess fat. Nonalcoholic steatohepatitis (NASH) is one type of NAFLD. Individuals with obesity and type 2 diabetes appear to be at greater risk of developing NAFLD. In recent years, NASH and NAFLD have been renamed MASH (metabolic dysfunction-associated steatohepatitis) and MASLD (metabolic dysfunction-associated steatotic liver disease), respectively.
Many innovative and scientifically intriguing therapeutic options are emerging for NASH. Clinical pharmacology considerations must be accounted for when developing treatments for these disorders. They are as follows.
Ensuring bench-to-bed translation
Preclinical models of NASH contain design and validation challenges and inadequate replication of human disease (1). Moreover, the necessity of liver histology predisposes the experiments to longer-term readouts. This makes translating research findings from animals to humans that much harder. Invariably, even shorter-term human experiments and translating to patient disease state is also a challenge.
Clinical and regulatory scientists accept and negotiate multiple biomarkers and endpoints for NASH. Early human experiments should include biomarker-rich sampling to understand the drug’s mechanism of action (target-specific biomarkers). This sampling will also aid in understanding the extent, duration, and nature of drug-target engagement.
The timings of liver biopsies limit longer trials in later phases of development. So, think about how to ensure sufficient translatability from early human to late human experiments. The increased emphasis on preventing cirrhosis in NASH has fueled translational models that measure the decline in disease progression.
How can you strengthen your translational objectives and reduce uncertainty in asset development?
First, design clinical trials that use an acceptable battery of endpoints. Second, supplement your clinical development plan with a cohesive model-informed development plan
Designing an appropriate hepatic impairment trial
Because small molecule drugs are routinely metabolized by the liver, a hepatic impairment study is typically considered a clinical pharmacology best practice. However, NASH drug development programs have one complicating feature. If NASH results in cirrhosis and subsequently, liver failure, then the consequences of disease progression on survival become critical.
The role of the hepatic impairment study is then a safety assessment as well. For this reason, design the hepatic impairment study to meet pharmacokinetic and safety objectives. A multiple-dose hepatic impairment study is a common scenario in this regard. Explore the work of Edwards et al (2) that persuasively explores these aspects.
Ensuring sufficient data rigor to assess longer-term exposure/response and exposure/safety
NASH exhibits slow disease progression (3). Therefore, the disease state’s impact on pharmacokinetics could change from the start of treatment to later in the maintenance phase. In addition, the emphasis on drug-related liver injury is a consideration for NASH.
Thus, set up the PK and pharmacodynamic (PD) sampling schedule in pivotal studies to address both short-term and long-term objectives. This would entail collecting PK samples from all subjects enrolled in a trial. Oftentimes, such sampling comes at a small logistical overhead. However, PK sampling of all subjects is invaluable.
Model-based solutions can help balance trial objectives and sampling. One example could be a balance between rich PK sampling in some subjects and sparse sampling in others. Common statistical methods can ensure the right balance. As trials are designed with adaptive components (whether dose and/or sample size), incorporating smart PK sampling choices can help optimize dosing for later phase trials and/or the drug label.
Building a physiologically significant pharmacokinetic model
There is likely a subtle interplay between the target tissue concentrations and those commonly sampled from the systemic central compartment. Often, systemic concentrations can explain compartmental pharmacokinetics. However, systemic concentrations can be misleading. Consider this aspect carefully early in the clinical program.
An example of this approach is borrowed from the work of Edwards et al (2). Their landmark work showed that while moderate and severe hepatic impairment significantly increased the systemic exposure of obeticholic acid, predicted liver concentrations did not. The liver is the site of pharmacological activity. Therefore, liver concentrations are more likely to be meaningful than plasma concentrations. Their results confirmed that the liver‐to‐plasma ratio differs between healthy and hepatically-impaired volunteers.
In summary, drug development for NASH is in a phase of scientific innovation. The translatability challenges present opportunities to bootstrap the clinical program with a rational choice of biomarkers and PK/PD approaches to consider the dose, dosing regimen, and target engagement. The regulatory frameworks in major markets present opportunities for the drug developer, as seen in a recent webcast (4).
Case Study: Rezdiffra (resmetirom) for NASH
In a landmark decision, the US FDA approved Madrigal Pharmaceuticals’ Rezdiffra (resmetirom) for treating adults with NASH with moderate to advanced fibrosis, along with diet and exercise.
Resmetirom has a novel mechanism of action. It is a partial agonist of the thyroid hormone receptor-beta (THR-β). Resmetirom produces >80% of the maximum response compared to triiodothyronine (T3), with an EC50 of 0.2 µM for THR-β activation.
THR-β is the major form of THR in the liver. Thus, stimulating THR-β in the liver reduces intrahepatic triglycerides. Resmetirom reduces liver fat content as measured by magnetic resonance imaging-protein density fat fraction (MRI-PDFF) or FibroScan controlled attenuation parameter (CAP) (Figure 1).
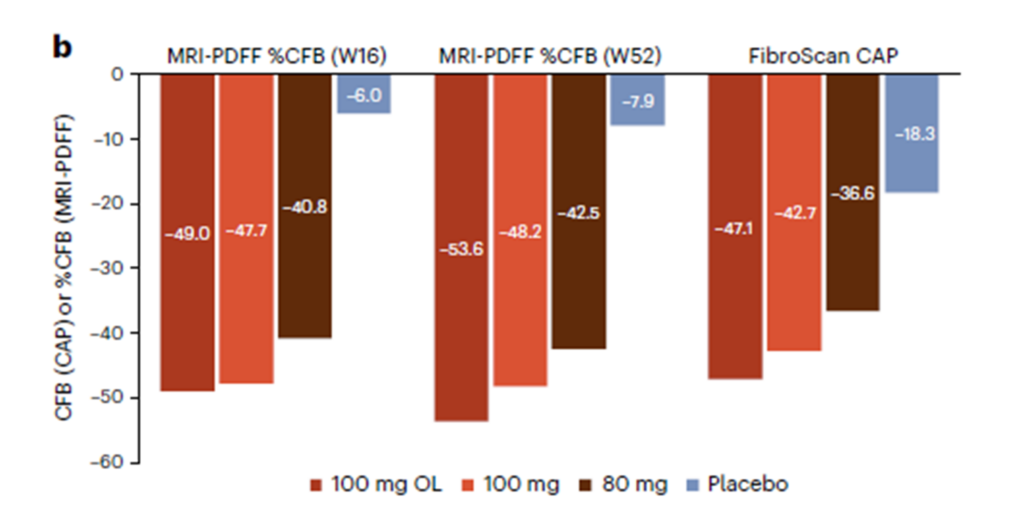
The FDA approved resmetirom under the accelerated approval pathway. Thus, the approval hinges on the weight of a surrogate or intermediate clinical endpoint that is reasonably likely to predict clinical benefit. Notably, the required study will assess clinical benefit after 54 months of treatment. Resmetirom also received Breakthrough Therapy, Fast Track, and Priority Review designations for NASH.
Interestingly, the hepatic impairment section of the package insert has the following text:

The surrogate endpoint measured the extent of liver inflammation and fibrosis. The accelerated approval pathway requires conversion to full approval. Therefore, the sponsor must conduct a post-approval study to demonstrate clinical benefit. For more details, please see the package insert.
Certara’s experts can help you build a model-informed clinical pharmacology program for NASH and other liver diseases. In addition, our CODEX MASH Database provides utility across the drug development continuum to inform critical decisions that determine not only a drug’s successful performance during a trial but also a drug’s profile within a competitive landscape.
To learn more about best practices for selecting and applying biomarkers within your drug development program, read this white paper.
This blog was originally posted on June 3, 2021. It was updated on August 23, 2024.
References
- Hansen HH, Feigh M, Veidal SS, et al. Mouse models of nonalcoholic steatohepatitis in preclinical drug development. Drug Discov Today. 2017;22:1707–18.
- Edwards JE et al. Modeling and Experimental Studies of Obeticholic Acid Exposure and the Impact of Cirrhosis Stage. Clin Trans Sci. 2016 Dec;9(6):328-336.
- Fazel Y, Koenig AB, Sayiner M, et al. Epidemiology and natural history of non-alcoholic fatty liver disease. Metabolism. 2016;65:1017–25.
- Regulatory Perspectives for Development of Drugs for Treatment of NASH. Jan 2021 (URL: https://www.fda.gov/drugs/news-events-human-drugs/regulatory-perspectives-development-drugs-treatment-nash-01292021-01292021).
- Harrison SA et al. Resmetirom for nonalcoholic fatty liver disease: a randomized, double-blind, placebo-controlled phase 3 trial. Nature Med. 2023 Nov;29(11):2919-2928.



