It is gratifying to see the continued uptake and adoption of tools for model-informed drug discovery and development since the early 2000s.1 Great advances have been made in modeling tools supporting target validation, safety, dose finding, and clinical trial optimization. The reporting of these tools and their applications in mainstream clinical science journals is influencing the way we conceptualize drug discovery and development. For the current COVID-19 pandemic, modeling tools are already helping to optimize and accelerate candidate therapeutics.2
Despite these advances, have we altered the way we address drug development questions? In particular, do we understand and espouse the science of quantitative decision making under uncertainty—that is, decision analysis—in the context of drug development? There are two aspects to this question: how we should make complex decisions under uncertainty and how do we to do so in practical ways?
Both aspects are addressed by various tools of decision analysis, such as multiattribute utility techniques3 and the Clinical Utility Index, which combines a drug candidate’s key attributes of efficacy, safety, and tolerability for dose optimization or for comparison across competing drugs.4-6 These tools are based on principles of management science and economics. Regardless of their origins, they change the dynamics of the conversation around rationality and choices in clinical development. Whether drug development decisions are around investment, prioritization, or dose-finding, the uncertainties behind successful development make most options risky. Complex decision problems with high levels of uncertainty are often left to individual judgment without the benefit of careful quantitative analysis (Figure 1).
There is nothing new about uncertainty in pharmaceutical development decisions. The difficulties of innovation and of anticipating medical needs decades into the future, as well as the uncertainty in the clinical relevance of discovery targets, have resulted in the development of more precedented targets, i.e., targets with some biological plausibility and/or validation. These present both investment and prioritization challenges to the decision maker. The current pandemic crisis can add another level of uncertainty: policy and regulatory uncertainty. While decision-analytic tools are well used in the areas of pharmaceutical availability, access, and pricing, there is less use in clinical development.
Multi-attribute utility analysis can help decision makers make transparent decisions under multiple criteria and uncertainty. For example, it can help drug developers decide whether to pause, terminate, or advance projects, or to select the projects with greatest expected returns on investment. For this to occur, decision makers need to absorb complex and imperfect information and synthesize an overall picture in transparent terms.
The Clinical Utility Index (CUI), an application of multiattribute utility theory7, is a tool to aid this synthesis. Figure 2 illustrates the combination of attributes, such as measures of efficacy and safety: the aggregate response utility is a function of response levels for individual attributes. These attributes can range from clinical endpoints or biomarker effects to physico-chemical compound properties, market factors, access, pricing, and costs. These variables can be distilled into an aggregate utility score for ranking and choice selection. Decision makers can test the sensitivity of the overall utility to the weights specifying each attribute’s contribution. Uncertainties are captured in probability distributions for the attribute levels, which induce a probability distribution on the aggregate CUI.
Such tools are components of a systematic decision-making process that includes structuring the decision problem (including objectives and alternatives to consider), quantifying the key uncertainties, iteratively refining the modeling with sensitivity analyses, and developing a commitment to implement the decision. The advantage of this process is that it offers a rational and transparent approach to making difficult trade-offs under uncertainty. It can reduce guesswork and advocacy not guided by quantitative processes when making complex strategic decisions. The process can be socialized within drug discovery, development, and regulatory teams wherever they may reside, in the pharmaceutical industry, academic laboratory, or government.
In addition to guiding pharmaceutical company decision making, integrated decision-analytic modeling can support policy making. For example, Kamal et al. (2017)8 assimilated various models generated during the course of clinical development of the antiviral oseltamivir, namely pharmacology, epidemiology, and health economics. This integrated framework allowed demonstration that oseltamivir at low and high doses would provide a meaningful reduction in the number of infected individuals, increase quality-adjusted life years, and enable cost savings under most influenza pandemic scenarios.
The ever-increasing complexity of information streams generated over the course of drug development programs and the disparate nature of data sources from externally generated studies present challenges for decision makers. In addition, the changing nature of the drug discovery process, as well as of regulation and pricing, underscore the need for a decision analytic framework to increase the rigor and transparency of the decision making process. This will lead to more careful consideration of alternatives, more quantitative consideration of uncertainties, and ultimately, higher quality decisions.
As innovation ecosystems and data sources continue to evolve, the challenge of making good choices and trade-offs calls increasingly for a rigorous framework, whether at the corporate or public policy level. The current pandemic is presenting drug developers and policy makers with many urgent choices in the development of candidate treatments and regimens, calling all the more for decision analysis and its practical tools for quantifying and comparing the options. By using decision analysis tools, we can accelerate drug development for COVID-19 and future pandemics.
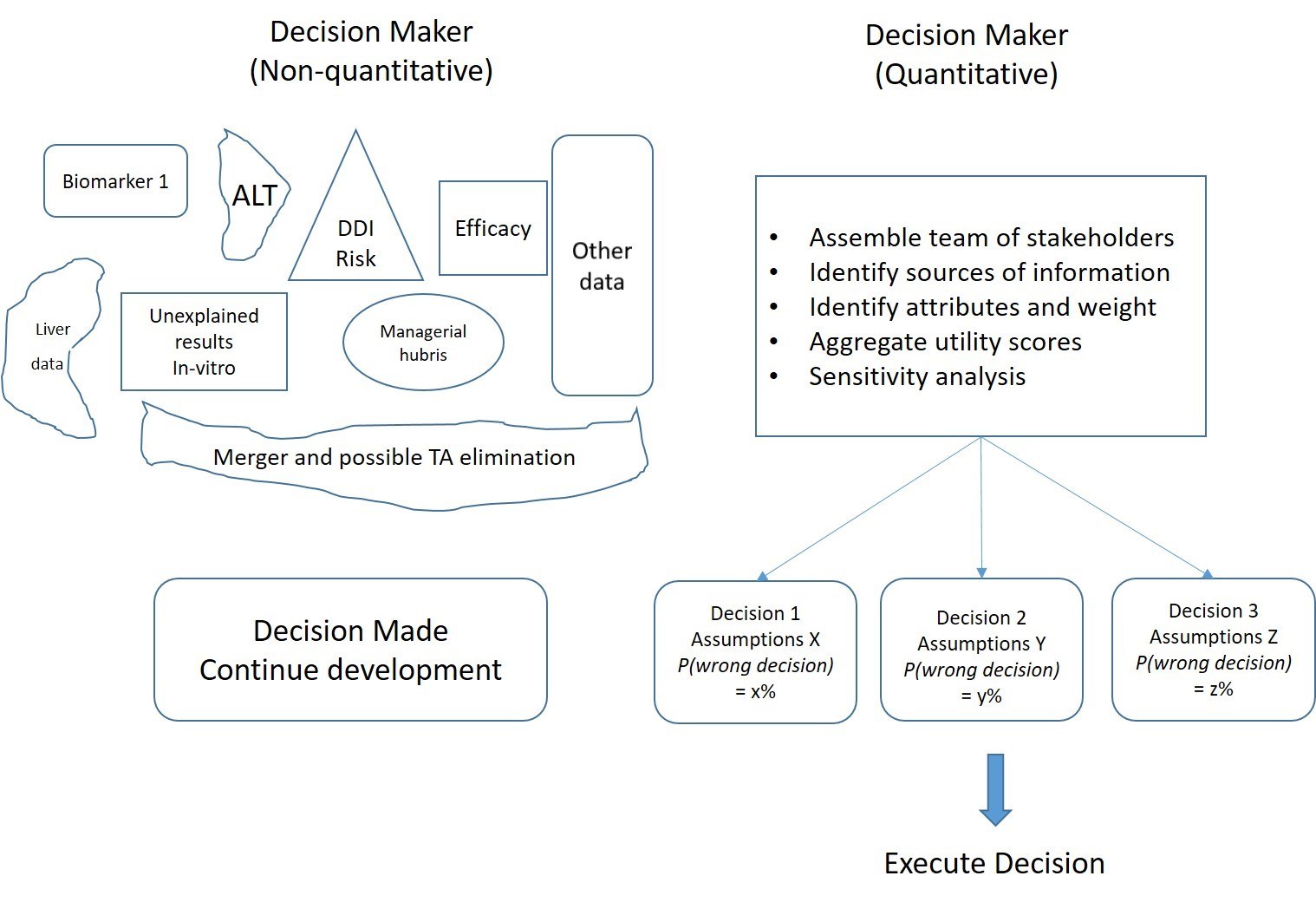
Figure 1: A simplistic view of approaches to decision-making about continuance of an asset development program
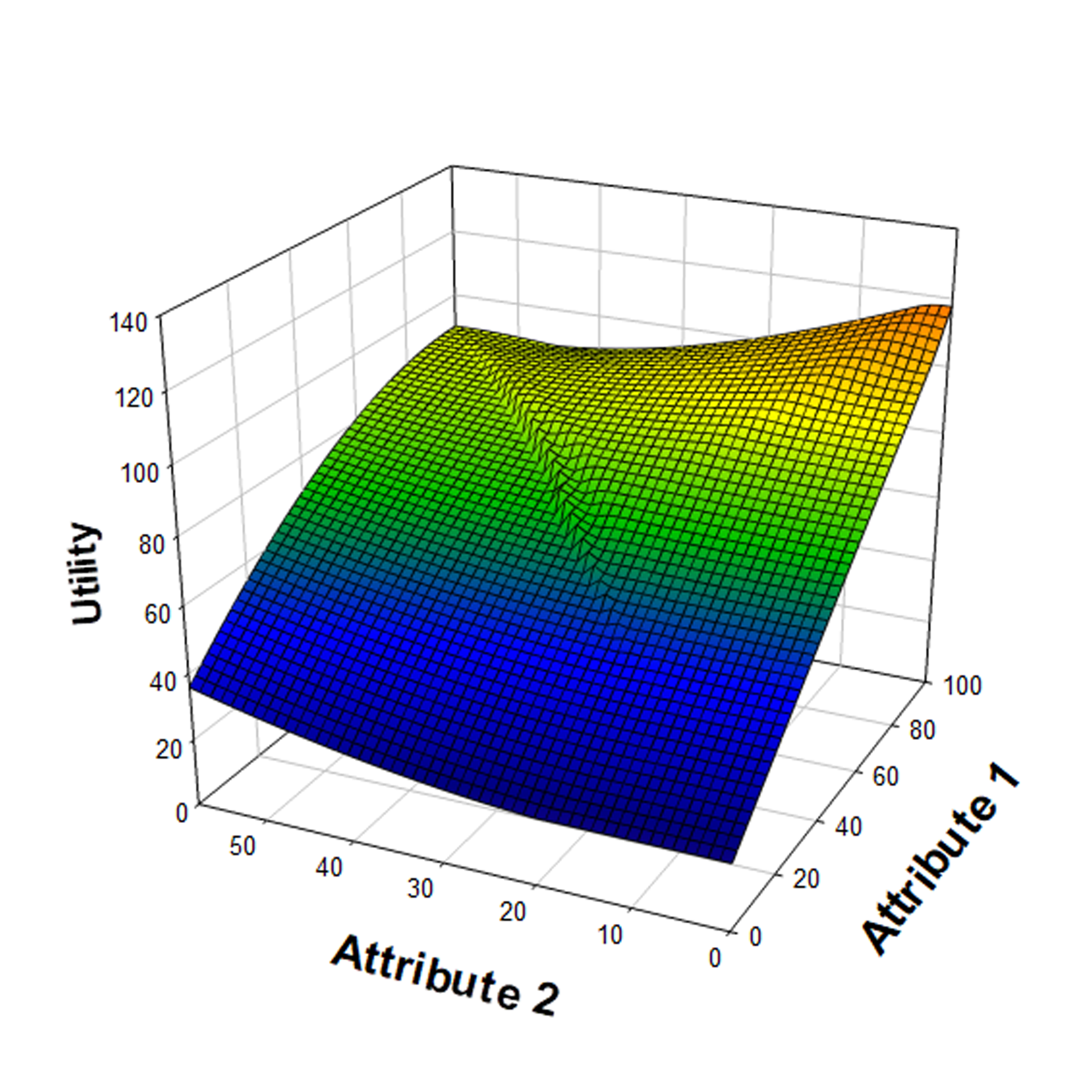
Figure 2: Visualization of a Clinical Utility Index aggregating response attributes. Typically, more than two attributes would be included. Efficacy-related attributes provide increase with dose, while safety and tolerability attributes decrease with dose.
References
- Krishna R. Quantitative clinical pharmacology (QCP): making paradigm shifts a reality. J Clin Pharmacol, 2006; 46: 966-967.
- Rayner C, Smith P, Hershberger K, Wesche D. Optimizing COVID-19 candidate therapeutics: Thinking Without Borders. Clin Transl Sci 202 Mar 25[Online ahead of print].
- Clemen RT, Reilly T. Making Hard Decisions: An Introduction to Decision Analysis. 2nd Duxbury Press, Belmont, CA, 1996.
- Poland B, Hodge LF, Khan AA, Clemen R, Wagner JA, Dykstra K, Krishna R. The clinical utility index as a practical multiattribute approach to drug development decisions. Clin Pharmacol Ther 2009;86(1):105-8.
- Zhu R, Poland B, Wada R, Liu Q, Musib L, Maslyar D, Cho E , Yu W, Ma H,, Jin J, Budha N. Exposure–Response-Based Product Profile-Driven Clinical Utility Index for Ipatasertib Dose Selection in Prostate Cancer. CPT Pharmacometrics Syst Pharmacol, 2019; 8(4):240-248.
- Khan AA, Perlstein I, Krishna R. The use of clinical utility assessments in early clinical development. In Meibohm B (Theme Editor), Quantitative Pharmacology, a Roadmap for Model-Based Development. AAPS Journal 2009;11(1):33-8.
- Keeney R, Raiffa H. Decisions with Multiple Objectives: Preferences and Value Tradeoffs. Wiley, New York; 1976.
- Kamal MA, Smith PF, Chaiyakunapruk N, Wu DBC, Pratoomsoot C, Lee KC, Yi Chong H, Nelson RE, Nieforth K, Dall G, Toovey S, Kong DCM, Kamauu A, Kirkpatrick CM, Rayner CR. Interdisciplinary pharmacometrics linking oseltamivir pharmacology, influenza epidemiology and health economics to inform antiviral use in pandemics. Br J Clin Pharmacol 2017; 83:1580–1594.
To learn more about this topic, please watch this webinar:


