January 17, 2025
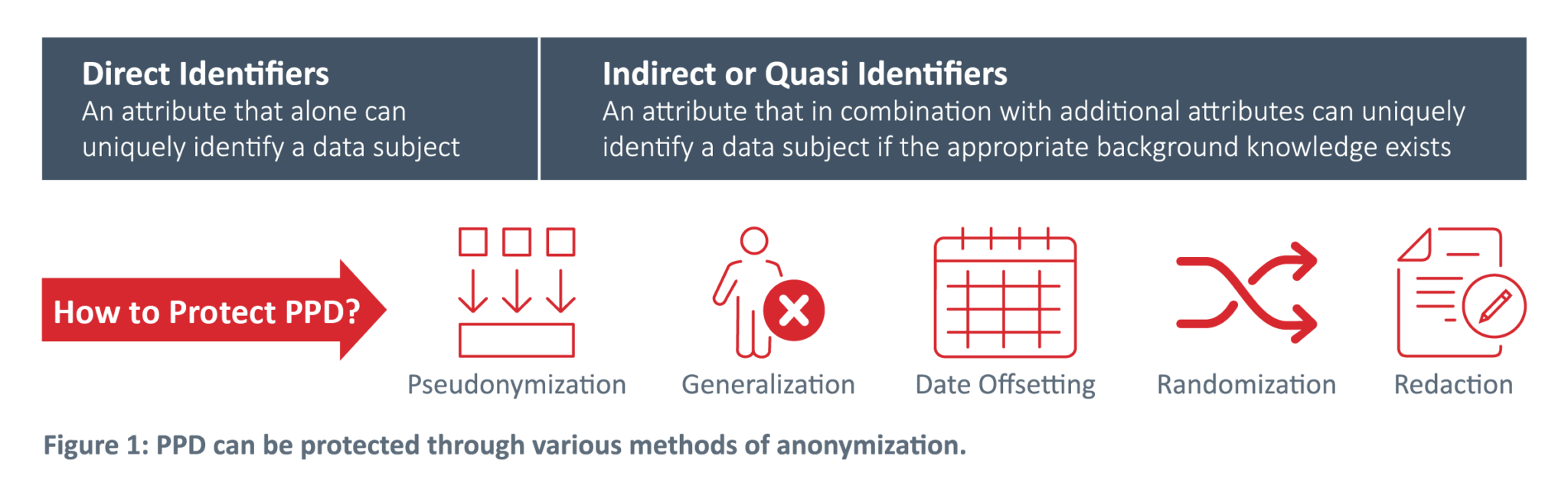

Clinical Trial Data Sharing Under EMA Policy 0070
Read our white paper to learn how to save time and resources while disclosing clinical trial data.

Senior Transparency Specialist
With a research background in neuroscience, and experience in health information technology, Honz Slipka has a thorough understanding of data analysis, regulatory standards, and best practices in the field of clinical data privacy. Drawing from his experiences, Honz is a champion of innovation, helping to lead the field of science, healthcare, and research into the modern age of technological efficiency, clinical transparency, and data utility.
This blog was originally published on October 5, 2022, and was updated on January 17, 2025.
Contact us





