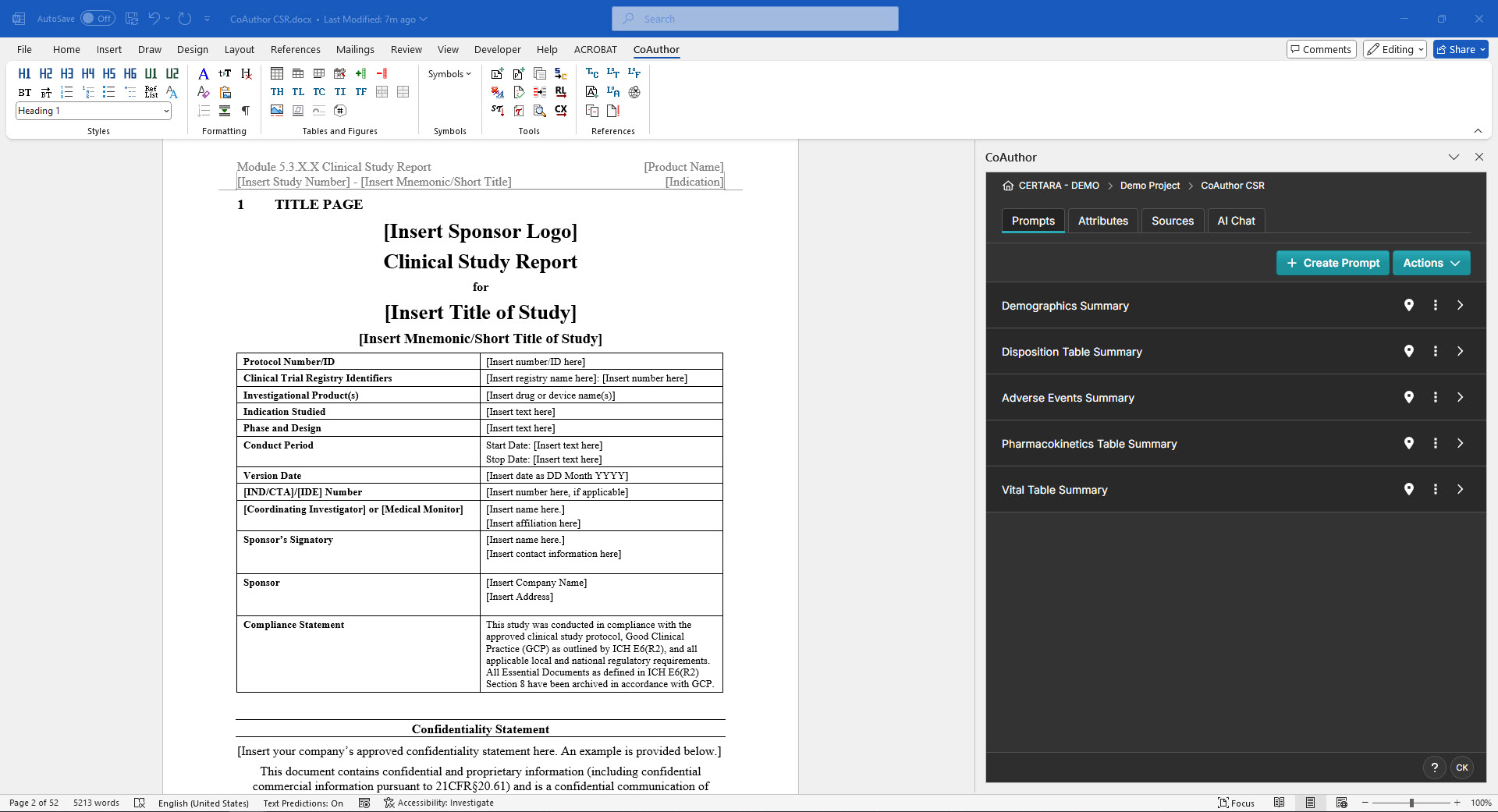
Discover how CoAuthor™ can streamline the drafting of regulatory documents directly in Microsoft Word. Combining a life science-specialized GenAI, structured content authoring, and a comprehensive eCTD template library, CoAuthor empowers regulatory writing teams to focus on contextualizing data and developing key messages. With built-in transparency, consistency, and collaboration tools, CoAuthor reduces submission timelines, enabling faster access to life-saving medicines for patients.
Developed by regulatory writers, for regulatory writers, CoAuthor simplifies the complexities of regulatory writing while ensuring safe and secure use of your data behind your organization-specific firewall.