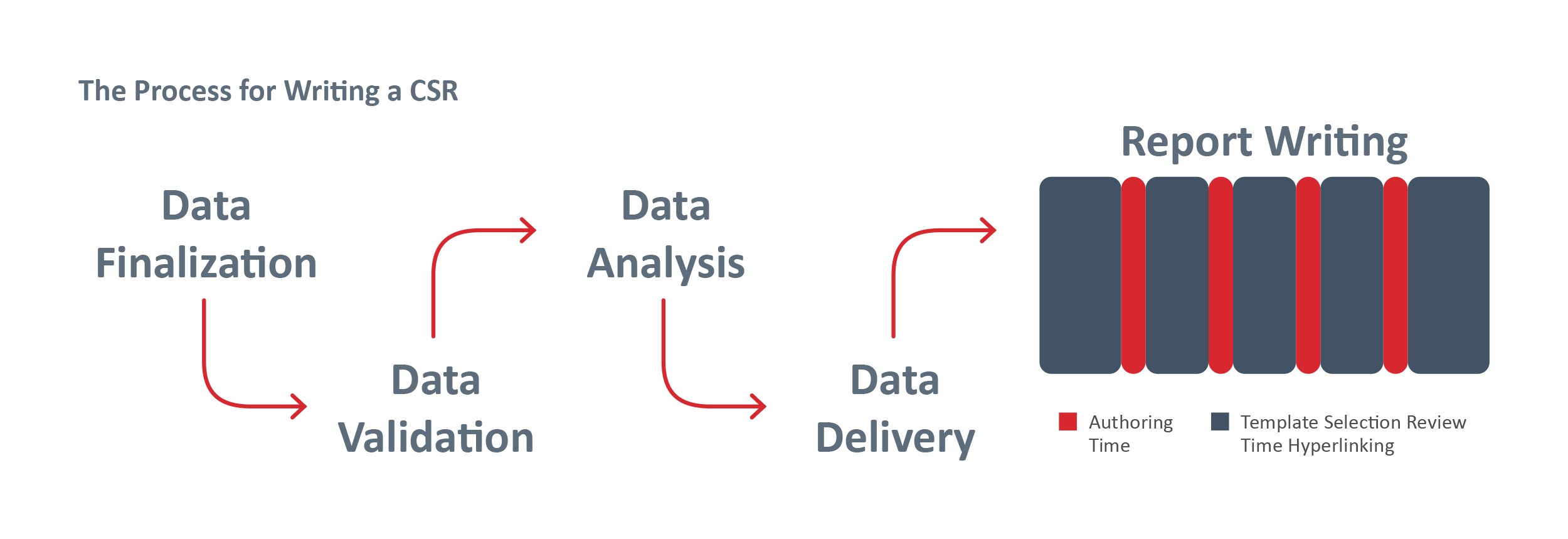
The demands of pharmaceutical regulatory document creation can be overwhelming—countless hours are spent compiling, reviewing, and formatting content. Even small mistakes in this critical process can cause costly delays in drug approvals, ultimately impacting patient care.
WHITE PAPER
Streamline Regulatory Writing with GenAI

About Certara
Certara is dedicated to transforming drug discovery and development for good. We harness the power of biosimulation, advanced analytics, and regulatory expertise to create a future where treatments reach patients faster and more efficiently.
From discovery to market access and commercial, we tailor solutions to meet our clients’ most pressing challenges. Through strategic leadership and advanced predictive technologies, Certara provides comprehensive solutions to optimize drug development processes, reduce risks, and improve outcomes. Our clients include more than 2,400 biopharmaceutical companies, academic institutions, and regulatory agencies across 70 countries.
© 2025 Certara. All Rights Reserved. | Privacy policy